Chapter 4: Q21E (page 714)
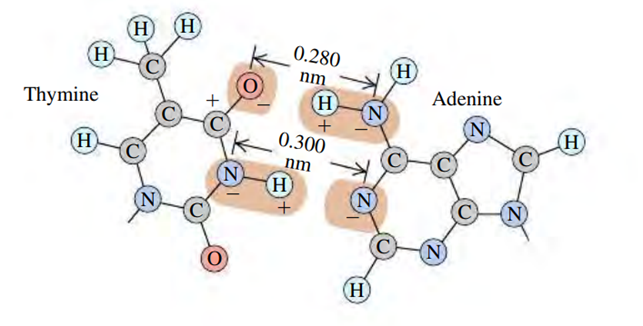
BIO Base Pairing in DNA, I. The two sides of the DNA double helix are connected by pairs of bases (adenine, thymine, cytosine, and guanine). Because of the geometric shape of these molecules, adenine bonds with thymine and cytosine bonds with guanine. Figure E21.21 shows the bonding of thymine and adenine. Each charge shown is, and the H—N distance is 0.110 nm. (a) Calculate the net force that thymine exerts on adenine. Is it attractive or repulsive? To keep the calculations fairly simple, yet reasonable, consider only the forces due to the O—H—N and the N—H—N combinations, assuming that these two combinations are parallel to each other. Remember, however, that in the O—H—N set, theexerts a force on both theand the, and likewise along the N—H—N set. (b) Calculate the force on the electron in the hydrogen atom, which is 0.0529 nm from the proton. Then compare the strength of the bonding force of the electron in hydrogen with the bonding force of the adenine–thymine molecules.
Short Answer
- Net force by thymine exerting on adenine is , and it is an attractive force.
- The force between electron and proton in the hydrogen atom is , and its ratio is 9.3

