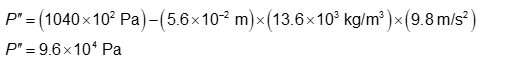Chapter 10: Q22P (page 260)
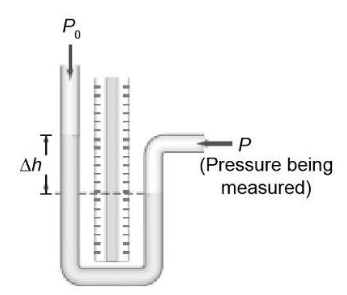
(II) An open-tube mercury manometer measures the pressure in an oxygen tank. When the atmospheric pressure is 1040 m bar, what is the absolute pressure (in Pa) in the tank if the height of the mercury in the open tube is (a) 18.5cm higher, (b) 5.6cm lower than the mercury in the tube connected to the tank? See Fig. 10–7a.
Short Answer
(a) The absolute pressure is 129 x 103 Pa.
(b) The absolute pressure is 9.6 x 104 Pa.