Chapter 1: Q29E (page 1)
Obtain the smoothness conditions at the boundaries between regions for the barrier (i.e., tunneling) case.
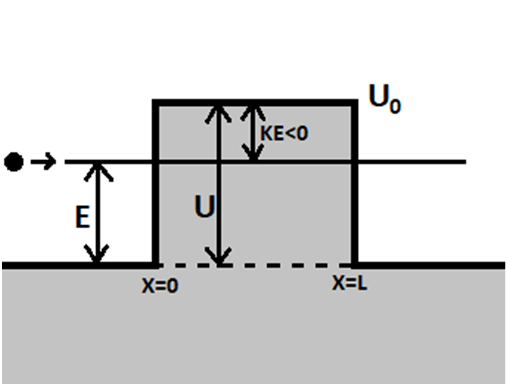
Short Answer
The four conditions that are boxed above are the smoothness condition for the given condition.
Learning Materials
Features
Discover
Chapter 1: Q29E (page 1)
Obtain the smoothness conditions at the boundaries between regions for the barrier (i.e., tunneling) case.

The four conditions that are boxed above are the smoothness condition for the given condition.
All the tools & learning materials you need for study success - in one app.
Get started for free
Calculate the probability that the electron in a hydrogen atom would be found within 30 degrees of the xy-plane, irrespective of radius, for (a) I=0 ,; (b) role="math" localid="1660014331933" and (c) . (d) As angular momentum increases, what happens to the orbits whose z-components of angular momentum are the maximum allowed?
Exercise 81 obtained formulas for hydrogen like atoms in which the nucleus is not assumed infinite, as in the chapter, but is of mass, whileis the mass of the orbiting negative charge. (a) What percentage error is introduced in the hydrogen ground-state energy by assuming that the proton is of infinite mass? (b) Deuterium is a form of hydrogen in which a neutron joins the proton in the nucleus, making the nucleus twice as massive. Taking nuclear mass into account, by what percent do the ground-state energies of hydrogen and deuterium differ?
Potassium-40 (Z=19 ,n=21) is a radioactive isotope that is rare but not unknown in nature. It is particularly interesting in that it lies along the curve of stability yet decays by both and -that is. in both directions away from the curve of stability. (a) Identify the daughter nuclei for both decays, (b) Many factors governing nuclear stability are discussed in the chapter (e.g., those in the semiempirical binding energy formula. magic numbers. and even numbers). Identify those that would argue only for decay. (c) Which factors argue only for decay? (d) Which argue for either decay?
A particle is subject to a potential energy that has an essentially infinitely high wall at the origin, like the infinite well, but for positive values of x is of the form U(x)= -b/ x, where b is a constant
(a) Sketch this potential energy.
(b) How much energy could a classical particle have and still be bound by such a potential energy?
(c) Add to your sketch a plot of E for a bound particle and indicate the outer classical tuning point (the inner being the origin).
(d) Assuming that a quantum-mechanical description is in order, sketch a plausible ground-state wave function, making sure that your function's second derivative is of the proper sign when U(x)is less than E and when it is greater.
The state –2p the state in which has most of its probability density along the z-axis, and so it is often referred to as a state. To allow its probability density to stick out in other ways and thus facilitate various kinds of molecular bonding with other atoms, an atomic electron may assume a wave function that is an algebraic combination of multiple wave functions open to it. One such “hybrid state” is the sum (Note: Because the Schrodinger equation is a linear differential equation, a sum of solutions with the same energy is a solution with that energy. Also, normalization constants may be ignored in the following questions.)
(a) Write this wave function and its probability density in terms of r, , and , (Use the Euler formula to simplify your result.)
(b) In which of the following ways does this state differ from its parts (i.e., and ) and from the 2pz state: Energy? Radial dependence of its probability density? Angular dependence of its probability density?
(c) This state is offer is often referred to as the . Why?
(d) How might we produce a state?
What do you think about this solution?
We value your feedback to improve our textbook solutions.