Chapter 1: Q11CQ (page 1)
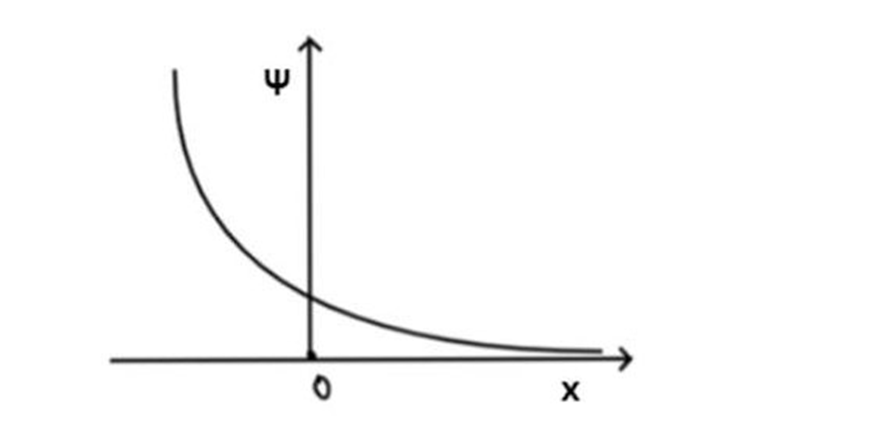
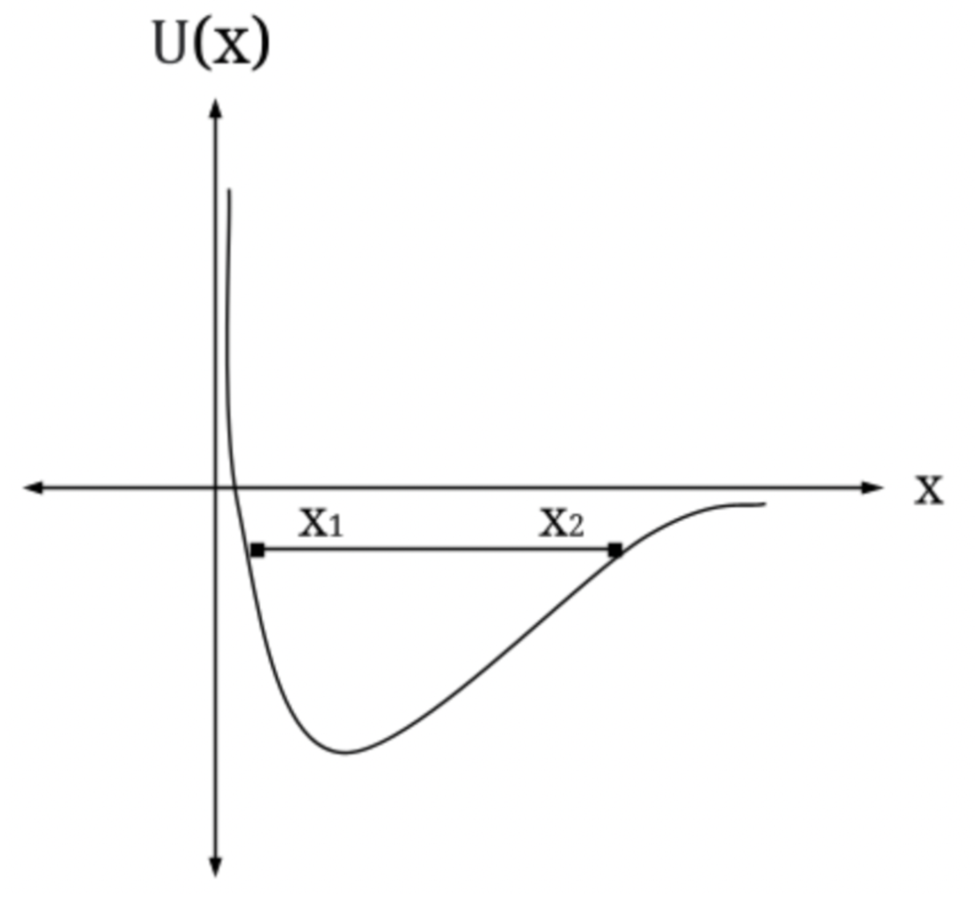
A particle is subject to a potential energy that has an essentially infinitely high wall at the origin, like the infinite well, but for positive values of x is of the form U(x)= -b/ x, where b is a constant
(a) Sketch this potential energy.
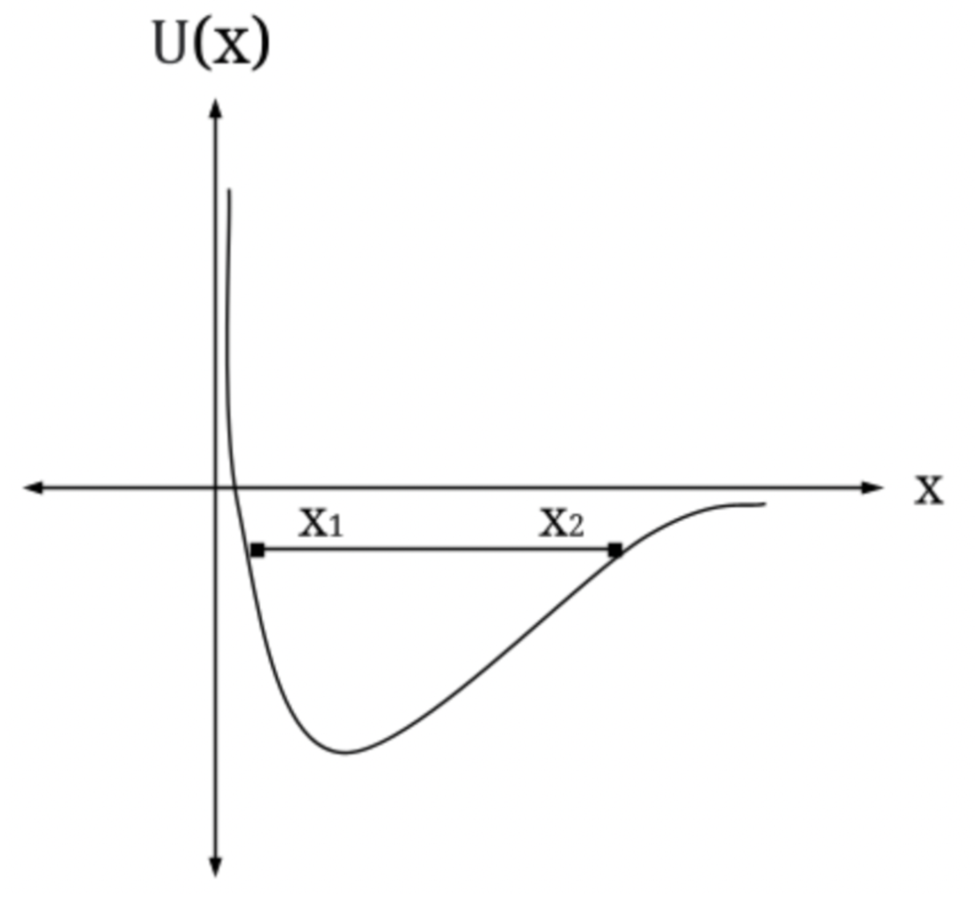
(b) How much energy could a classical particle have and still be bound by such a potential energy?
(c) Add to your sketch a plot of E for a bound particle and indicate the outer classical tuning point (the inner being the origin).
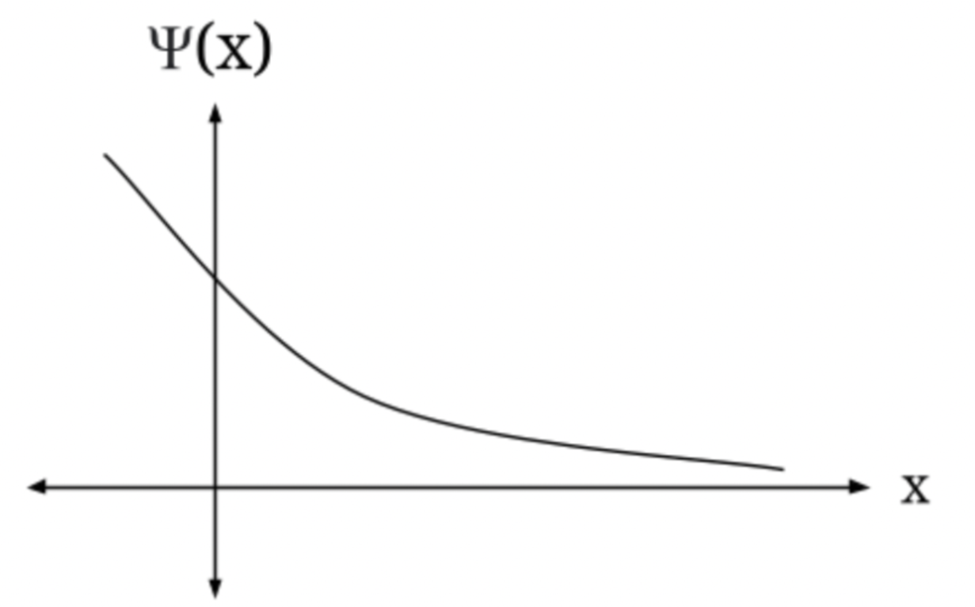
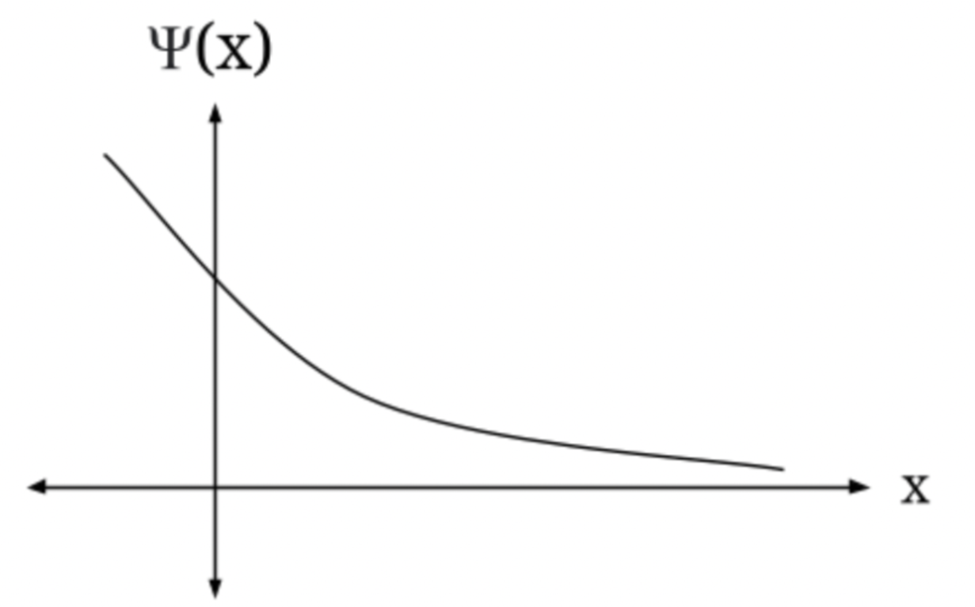
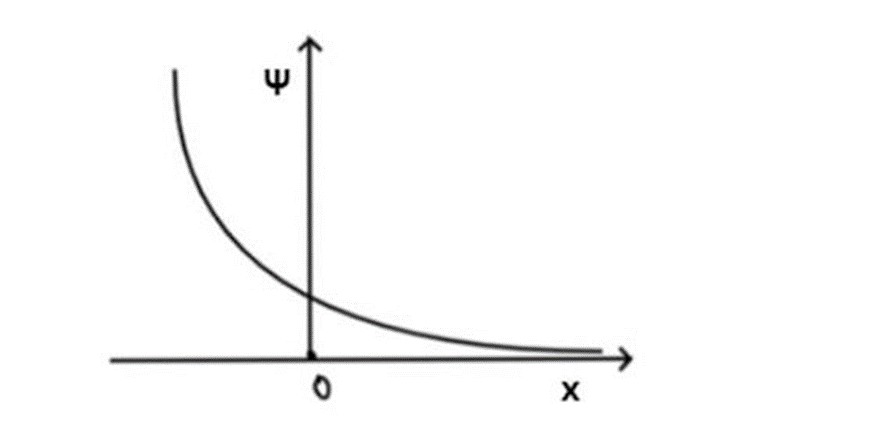
(d) Assuming that a quantum-mechanical description is in order, sketch a plausible ground-state wave function, making sure that your function's second derivative is of the proper sign when U(x)is less than E and when it is greater.
Short Answer
(a)The plot obtained is as follows,
(b) The energy is equal to E = 0 when the particle is free and E 0 to be still bounded by the given potential energy
(c) the graph
(d) the graph of wave function