Chapter 5: Q95CE (page 193)
A bound particle of massdescribed by the wave function
What is the most probable location at which to find the particle?
Short Answer
The most probably location to find the particle is at.
Learning Materials
Features
Discover
Chapter 5: Q95CE (page 193)
A bound particle of massdescribed by the wave function
What is the most probable location at which to find the particle?
The most probably location to find the particle is at.
All the tools & learning materials you need for study success - in one app.
Get started for free
Determine the expectation value of the position of a harmonic oscillator in its ground state.
Explain to your friend, who is skeptical about energy quantization, the simple evidence provided by distinct colors you see when you hold a CD (serving as grating) near a fluorescent light. It may be helpful to contrast this evidence with the spectrum produced by an incandescent light, which relies on heating to produce a rather nonspecific blackbody spectrum.
The figure shows a potential energy function.
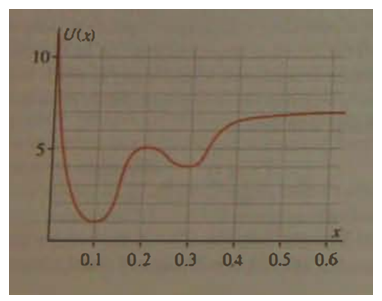
(a) How much energy could a classical particle have and still be bound?
(b) Where would an unbound particle have its maximum kinetic energy?
(c) For what range of energies might a classical particle be bound in either of two different regions?
(d) Do you think that a quantum mechanical particle with energy in the range referred to in part?
(e) Would be bound in one region or the other? Explain.
Show that the uncertainty in the momentum of a ground state harmonic oscillator is .
Show that the uncertainty in a particle’s position in an infinite well in the general case of arbitrary is given by
Discuss the dependence. In what circumstance does it agree with the classical uncertainty of discussed in Exercise 55?
What do you think about this solution?
We value your feedback to improve our textbook solutions.