Chapter 5: Q59E (page 191)
Determine the expectation value of the position of a harmonic oscillator in its ground state.
Short Answer
The expectation value of harmonic oscillator in ground state is 0.
Learning Materials
Features
Discover
Chapter 5: Q59E (page 191)
Determine the expectation value of the position of a harmonic oscillator in its ground state.
The expectation value of harmonic oscillator in ground state is 0.
All the tools & learning materials you need for study success - in one app.
Get started for free
The harmonic oscillator potential energy is proportional to , and the energy levels are equally spaced:
. The energy levels in the infinite well become farther apart as energy increases: .Because the functionis 0 forand infinitely large for. the infinite well potential energy may be thought of as proportional to .
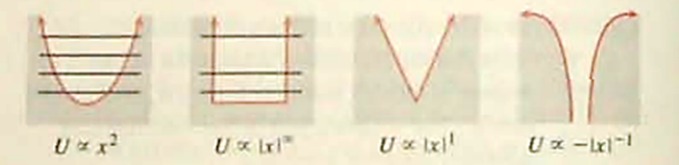
How would you expect energy levels to be spaced in a potential well that is (a) proportional to and (b) proportional to ? For the harmonic oscillator and infinite well. the number of bound-state energies is infinite, and arbitrarily large bound-state energies are possible. Are these characteristics shared (c) by the well and (d) by thewell? V
Verify that solution (5-19) satisfies the Schrodinger equation in form (5.18).
In several bound systems, the quantum-mechanically allowed energies depend on a single quantum number we found in section 5.5 that the energy levels in an infinite well are given by, whereandis a constant. (Actually, we known whatis but it would only distract us here.) section 5.7 showed that for a harmonic oscillator, they are, where(using anwith n strictly positive is equivalent towith n non negative.) finally, for a hydrogen atom, a bound system that we study in chapter 7,, whereconsider particles making downwards transition between the quantized energy levels, each transition producing a photon, for each of these three systems, is there a minimum photon wavelength? A maximum ? it might be helpful to make sketches of the relative heights of the energy levels in each case.
The deeper the finite well, the more state it holds. In fact, a new state, the, is added when the well’s depthreaches. (a) Argue that this should be the case based only on, the shape of the wave inside, and the degree of penetration of the classically forbidden region expected for a state whose energy E is only negligibly below. (b) How many states would be found up to this same “height” in an infinite well.
What is the product of uncertainties determined in Exercise 60 and 61? Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.