Chapter 5: Q47E (page 189)
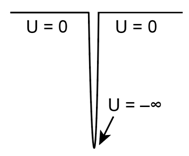
Consider the delta well potential energy:
Although not completely realistic, this potential energy is often a convenient approximation to a verystrong, verynarrow attractive potential energy well. It has only one allowed bound-state wave function, and because the top of the well is defined as U = 0, the corresponding bound-state energy is negative. Call its value -E0.
(a) Applying the usual arguments and required continuity conditions (need it be smooth?), show that the wave function is given by
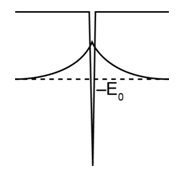
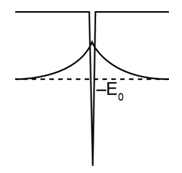
(b) Sketch and U(x) on the same diagram. Does this wave function exhibit the expected behavior in the classically forbidden region?
Short Answer
(a) It is verified that the wave function for the delta well potential is given by
(b) The plot of the wave function is given below. It exhibits expected behavior in the classically forbidden region.