Chapter 5: Q15CQ (page 186)
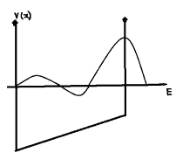
Consider a particle bound in a infinite well, where the potential inside is not constant but a linearly varying function. Suppose the particle is in a fairly high energy state, so that its wave function stretches across the entire well; that is isn’t caught in the “low spot”. Decide how ,if at all, its wavelength should vary. Then sketch a plausible wave function.
Short Answer
The graph is plotted with energy and potential inside a potential well.