Chapter 5: Q 9CQ (page 186)
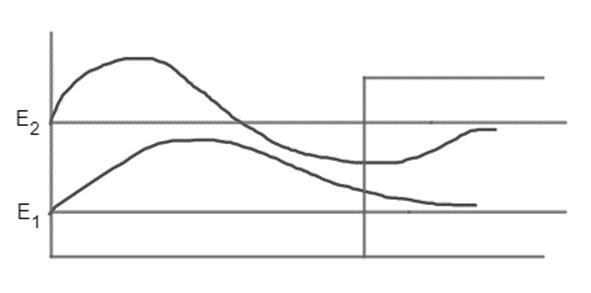
A half-infinite well has an infinitely high wall at the origin and one of finite height U0 at x= L . Like the finite well, the number of allowed states is limited. Assume that it has two states, of energy E1 and E2 , where E2 is not much below U0. Make a sketch of the potential energy, then add plausible sketches of the two allowed wave functions on separate horizontal axes whose heights are E1 and E2 .
Short Answer
E1 and E2 are two states of energy in a half-infinite square well. At a distance of L from the left wall, the right "step" has a height of U0 .