Chapter 19: Q68P (page 581)
In an interstellar gas cloud at,the pressure is. Assuming that the molecular diameters of the gases in the cloud are all, what is their mean free path?
Short Answer
The gas cloud has a mean free path equal to
Learning Materials
Features
Discover
Chapter 19: Q68P (page 581)
In an interstellar gas cloud at,the pressure is. Assuming that the molecular diameters of the gases in the cloud are all, what is their mean free path?
The gas cloud has a mean free path equal to
All the tools & learning materials you need for study success - in one app.
Get started for free
The temperature ofof a gas with is to be raised . If the process is at constant volume, what are (a) the energy transferred as heat Q, (b) the work W done by the gas, (c) the change in internal energy of the gas, and (d) the change in the total translational kinetic energy? If the process is at constant pressure, what are (e) Q, (f) W, (g) , and (h) ? If the process is adiabatic, what are (i) Q, (j) W, (k) , and (l)?
Does the temperature of an ideal gas increase, decrease, or stay the same during (a) an isothermal expansion, (b) an expansion at constant pressure, (c) an adiabatic expansion, and (d) an increase in pressure at constant volume?
Determine the average value of the translational kinetic energy of the molecules of an ideal gas at
a.
b.
What is the translational kinetic energy per mole of an ideal gas at
c.
d.
The temperature ofofan ideal diatomic gas is increased by without the pressure of the gas changing. The molecules in the gas rotate but do not oscillate.
a) How much energy is transferred to the gas as heat?
b) What is the change in the internal energy of the gas?
c) How much work is done by the gas?
d) By how much does the rotational kinetic energy of the gas increase?
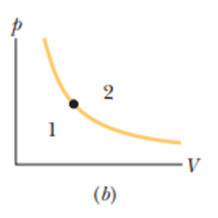
The dot in Figre 19-18bpresents the initial state of a gas, and the isotherm through the dot divides the p-V diagram into regions 1 and 2. For the following processes, determine whether the change in the internal energy of the gas is positive, negative, or zero: (a) the gas moves up along the isotherm, (b) it moves down along the isotherm, (c) it moves to anywhere in region, and (d) it moves to anywhere in region.
What do you think about this solution?
We value your feedback to improve our textbook solutions.