Chapter 19: Q42P (page 579)
What is the internal energy ofof an ideal monoatomic gas at?
Short Answer
The internal energy of of an ideal monoatomic gas at is .
Learning Materials
Features
Discover
Chapter 19: Q42P (page 579)
What is the internal energy ofof an ideal monoatomic gas at?
The internal energy of of an ideal monoatomic gas at is .
All the tools & learning materials you need for study success - in one app.
Get started for free
Suppose 1.00Lof a gas with, initially at 273K and1.00atm is suddenly compressed adiabatically to half its initial volume. Find
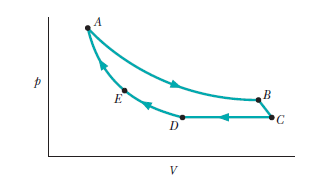
Question: Figureshows a cycle consisting of five paths: AB is isothermal at 300K, BC is adiabatic with , CD is at a constant pressure of, DE is isothermal, and EA is adiabatic with a change in internal energy of . What is the change in internal energy of the gas along path CD?
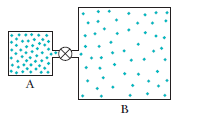
Question: Container A in figure holds an ideal gas at a pressure of and a temperature of 300 kIt is connected by a thin tube (and a closed valve) to container B, with four times the volume of A. Container B holds the same ideal gas at a pressure of and a temperature of 400 k. The valve is opened to allow the pressures to equalize, but the temperature of each container is maintained. What then is the pressure?
The temperature ofof a gas with is to be raised . If the process is at constant volume, what are (a) the energy transferred as heat Q, (b) the work W done by the gas, (c) the change in internal energy of the gas, and (d) the change in the total translational kinetic energy? If the process is at constant pressure, what are (e) Q, (f) W, (g) , and (h) ? If the process is adiabatic, what are (i) Q, (j) W, (k) , and (l)?
The speeds of 10molecules are
What do you think about this solution?
We value your feedback to improve our textbook solutions.