Chapter 19: Q2P (page 577)
Gold has a molar mass of ,
(a) How many moles of gold are in a sample ofpure gold?
(b) How many atoms are in the sample?
Short Answer
(a) 0.0127 mol of gold is in a sample of pure gold.
(b) There are atoms in the sample.
Learning Materials
Features
Discover
Chapter 19: Q2P (page 577)
Gold has a molar mass of ,
(a) How many moles of gold are in a sample ofpure gold?
(b) How many atoms are in the sample?
(a) 0.0127 mol of gold is in a sample of pure gold.
(b) There are atoms in the sample.
All the tools & learning materials you need for study success - in one app.
Get started for free
In a bottle of champagne, the pocket of gas (primarily carbon dioxide) between the liquid and the cork is at a pressure of.When the cork is pulled from the bottle, the gas undergoes an adiabatic expansion until its pressure matches the ambient air pressure of.Assume that the ratio of the molar specific heats is. If the gas has initial temperature, what is the temperature at the end of the adiabatic expansion?
For four situations for an ideal gas, the table gives the energy transferred to or from the gas as heat Qand either the work W done by the gas or the work done on the gas, all in joules. Rank the four situations in terms of the temperature change of the gas, most positive first.
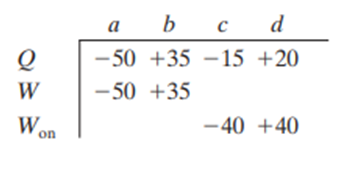
(a) Compute the RMS speed of a nitrogen molecule at 20.0.The molar mass of nitrogen molecules (N2) is given in Table. At what temperatures will the rms speed be (b) half that value and (c) twice that value?
Compute
At what temperature do atoms of helium gas have the same rms speed as molecules of hydrogen gas at ?
What do you think about this solution?
We value your feedback to improve our textbook solutions.