Chapter 18: Q88P (page 547)
A steel rod atis bolted at both ends and then cooled. At what temperature will it rupture? Use Table 12-1.
Short Answer
The temperature at which the steel rod will rupture is .
Learning Materials
Features
Discover
Chapter 18: Q88P (page 547)
A steel rod atis bolted at both ends and then cooled. At what temperature will it rupture? Use Table 12-1.
The temperature at which the steel rod will rupture is .
All the tools & learning materials you need for study success - in one app.
Get started for free
What mass of steam at must be mixed with of ice at its melting point, in a thermally insulated container, to produce liquid water at ?
Question: Suppose that on a linear temperature scale X, water boils at and freezes at. What is a temperature ofon the X scale? (Approximate water’s boiling point as 373K.)
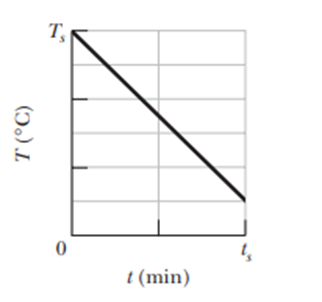
Question: A0.300 kg sample is placed in a cooling apparatus that removes energy as heat at a constant rate of 2.81 W . Figure 18-52 gives the temperature Tof the sample versus time t.The temperature scale is set by and the time scale is set by .What is the specific heat of the sample?
In a solar water heater, energy from the Sun is gathered by water that circulates through tubes in a rooftop collector. The solar radiation enters the collector through a transparent cover and warms the water in the tubes; this water is pumped into a holding tank. Assume that the efficiency of the overall system is (that is, of the incident solar energy is lost from the system).What collector area is necessary to raise the temperature of of water in the tank from to in when the intensity of incident sunlight is ?
Samples A and B are at different initial temperatures when they are placed in a thermally insulated container and allowed to come to thermal equilibrium. Figure a gives their temperatures T versus time t. Sample A has a mass of; sample B has a mass of . Figure b is a general plot for the material of sample B. It shows the temperature change that the material undergoes when energy is transferred to it as heat Q. The changeis plotted versus the energy Q per unit mass of the material, and the scale of the vertical axis is set by .What is the specific heat of sample A?
What do you think about this solution?
We value your feedback to improve our textbook solutions.