Chapter 18: Q38P (page 542)
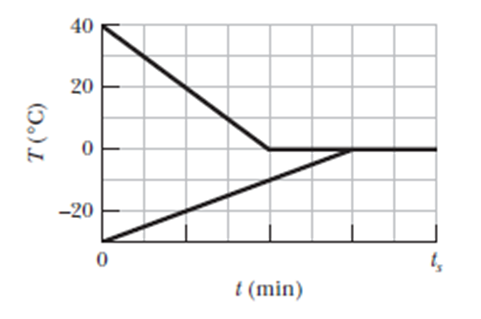
A 0.530 kgsample of liquid water and a sample of ice are placed in a thermally insulated container. The container also contains a device that transfers energy as heat from the liquid water to the ice at a constant rate P, until thermal equilibrium is reached. The temperatures Tof the liquid water and the ice are given in Figure as functions of time t; the horizontal scale is set by(a) What is rate P? (b) What is the initial mass of the ice in the container? (c) When thermal equilibrium is reached, what is the mass of the ice produced in this process?
Short Answer
- The rate P is 37 W
- The initial mass of the ice in the container is 2.0 kg
- The mass of the ice produced in the process when thermal equilibrium is reached is 0.13 kg