Chapter 18: Temperature, Heat, and the First Law of Thermodynamics
Q19P
A vertical glass tube of length is half filled with a liquid at. How much will the height of the liquid column change when the tube and liquid are heated to? Use coefficients
Q1P
Question: Suppose the temperature of a gas iswhen it is at the boiling point of water. What then is the limiting value of the ratio of the pressure of the gas at that boiling point to its pressure at the triple point of water? (Assume the volume of the gas is the same at both temperatures).
Q1Q
The initial length L, change in temperature , and change in length of four rods are given in the following table. Rank the rods according to their coefficients of thermal expansion, greatest first.
| Rod L (m) |
Q20P
In a certain experiment, a small radioactive source must move at selected, extremely slow speeds. This motion is accomplished by fastening the source to one end of an aluminum rod and heating the central section of the rod in a controlled way. If the effective heated section of the rod in Figure has length , at what constant rate must the temperature of the rod be changed if the source is to move at a constant speed of ?
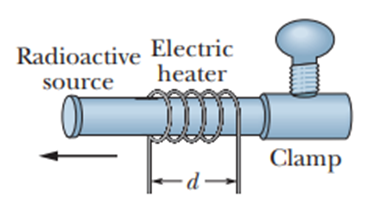
Q21P
As a result of a temperature rise of , a bar with a crack at its center buckles upward (Figure). If the fixed distance and the coefficient of linear expansion of the bar is , find the rise x of the center.
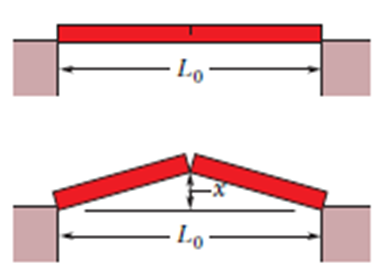
Q22P
One way to keep the contents of a garage from becoming too cold on a night when a severe subfreezing temperature is forecast is to put a tub of water in the garage. If the mass of the water is and its initial temperature is, (a) how much energy must the water transfer to its surroundings in order to freeze completely and (b) what is the lowest possible temperature of the water and its surroundings until that happens?
Q23P
A small electric immersion heater is used to heat of water for a cup of instant coffee. The heater is labeled “” (it converts electrical energy to thermal energy at this rate). Calculate the time required to bring all this water from, ignoring any heat losses.
Q24P
A certain substance has a mass per mole of 50.0 g/mol. When 314 J is added as heat to a 30.0 g sample, the sample’s temperature rises from 25.0°C to 45.0°C. (a) What is the specific heat and (b) What is the molar specific heat of this substance? (c) How many moles are in the sample?
Q25P
A certain diet doctor encourages people to diet by drinking ice water. His theory is that the body must burn off enough fat to raise the temperature of the water from to the body temperature of. How many liters of ice water would have to be consumed to burn off(about 1 lb) of fat, assuming that burning this much fat requiresbe transferred to the ice water? Why is it not advisable to follow this diet? (. The density of water is.)
Q26P
What mass of butter, which has a usable energy content ofwould be equivalent to the change in gravitational potential energy of aman who ascends from sea level to the top of Mt. Everest, at elevation? Assume that the average g for the ascent is.
