Chapter 38: Q6P (page 1181)
What is the photon energy for yellow light from a highway sodium lamp at a wavelength of 589 nm?
Short Answer
The photon energy of the light is 2.1 eV.
Learning Materials
Features
Discover
Chapter 38: Q6P (page 1181)
What is the photon energy for yellow light from a highway sodium lamp at a wavelength of 589 nm?
The photon energy of the light is 2.1 eV.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: An electron moves through a region of the uniform electric potential of with a (total) energy of . What are its (a) kinetic energy (in electron-volts), (b) momentum, (c) speed, (d) de Broglie wavelength, and (e) angular wave number?
What is the wavelength of (a) a photon with energy , (b) an electron with energy , (c) a photon of energy , and (d) an electron with energy ?
Gamma rays of photon energy 0.511 MeV are directed vonto an aluminium target and are scattered in various directions by loosely bound electrons there.
(a) What is the wavelength of the incident gamma rays?
(b) What is the wavelength of gamma rays scattered at to the incident beam?
(c) What is the photon energy of the rays scattered in this direction?
Show that when a photon of energy Eis scattered from a free electron at rest, the maximum kinetic energy of the recoiling electron is given by
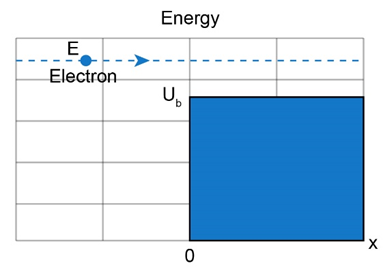
Question: For the arrangement of Figs. and , electrons in the incident beam in region 1 have energy and the potential step has a height of . What is the angular wave number in (a) region 1 and (b) region 2 ? (c) What is the reflection coefficient? (d) If the incident beam sends electrons against the potential step, approximately how many will be reflected?
Fig 38-14
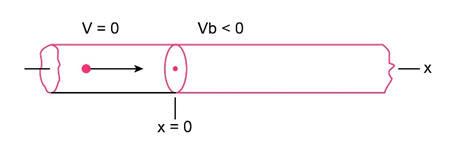
Fig 38-15
What do you think about this solution?
We value your feedback to improve our textbook solutions.