Chapter 38: Q 71P (page 1184)
Question: For the arrangement of Figs. and , electrons in the incident beam in region 1 have energy and the potential step has a height of . What is the angular wave number in (a) region 1 and (b) region 2 ? (c) What is the reflection coefficient? (d) If the incident beam sends electrons against the potential step, approximately how many will be reflected?
Fig 38-14
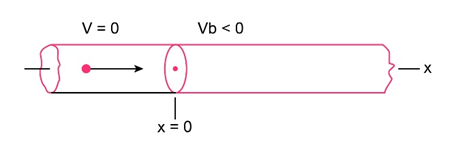
Fig 38-15
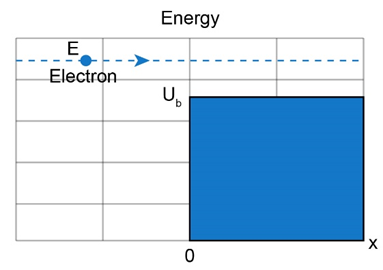
Short Answer
- The angular wave number of region is .
- The angular wave number of region is .
- The reflection coefficient is .
- The number of reflected electrons is .

