Chapter 38: Q2P (page 1181)
How fast must an electron move to have a kinetic energy equal to the photon energy of sodium light at wavelength 590 nm?
Short Answer
The speed of the electron is .
Learning Materials
Features
Discover
Chapter 38: Q2P (page 1181)
How fast must an electron move to have a kinetic energy equal to the photon energy of sodium light at wavelength 590 nm?
The speed of the electron is .
All the tools & learning materials you need for study success - in one app.
Get started for free
X rays of wavelength 0.0100 nm are directed in the positive direction of an axis onto a target containing loosely bound electrons. For Compton scattering from one of those electrons, at an angle of ,what are (a) the Compton shift, (b) the corresponding change in photon energy, (c) the kinetic energy of the recoiling electron, and (d) the angle between the positive direction of the axis and the electron’s direction of motion?
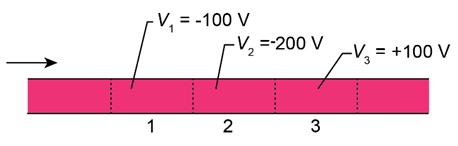
Figure 38-24 shows an electron moving through several regions where uniform electric potentials V have been set up. Rank the three regions according to the de Broglie wavelength of the electron there, greatest first.
The meter was once defined as 1650763.73 wavelengths of the orange light emitted by a source containing krypton-86 atoms. What is the photon energy of that light?
Using the classical equations for momentum and kinetic energy, show that an electron’s de Broglie wavelength in nanometres can be written as, in which Kis the electron’s kinetic energy in electron-volts.
Neutrons in thermal equilibrium with matter have an average kinetic energy of , where is the Boltzmann constant and T, which may be taken to be , is the temperature of the environment of the neutrons. (a) What is the average kinetic energy of such a neutron? (b) What is the corresponding de Broglie wavelength?
What do you think about this solution?
We value your feedback to improve our textbook solutions.