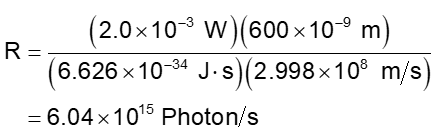The power output is given by
P = RE ….. (1)
Now, Energy is given by,
Here, h is the Plank’s constant, c is the speed of light and is the wavelength.
Substitute localid="1663150706210" for E into equation (1)
localid="1663150711000"
Solving the above equation and finding the value of R.
localid="1663150715861"
Consider the given data below.
The wavelength, localid="1663150719899"
The power, localid="1663150723665"
Plank’s constant, localid="1663150727678"
Speed of light, localid="1663150733098"
Substitute these numerical values
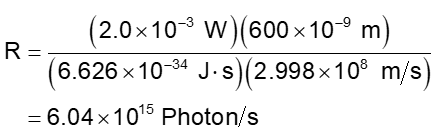
Now, the fractional efficiency of Cesium surface is
Therefore, the rate of photons that actually cause photoelectric emissions is
Now, the current is equal to the rate of electrons multiplied by the charge of an electron
Hence, the current of electrons ejected from the surface is