Chapter 38: Q12Q (page 1181)
An electron and a proton have the same kinetic energy. Which has the greater de Broglie wavelength?
Short Answer
The electron has greater de Broglie wavelength than proton.
Learning Materials
Features
Discover
Chapter 38: Q12Q (page 1181)
An electron and a proton have the same kinetic energy. Which has the greater de Broglie wavelength?
The electron has greater de Broglie wavelength than proton.
All the tools & learning materials you need for study success - in one app.
Get started for free
(a) In Me V/c, what is the magnitude of momentum associated with a photon having an energy equal to the electron rest energy? What are the (b) wavelength and (c) frequency of the corresponding radiation?
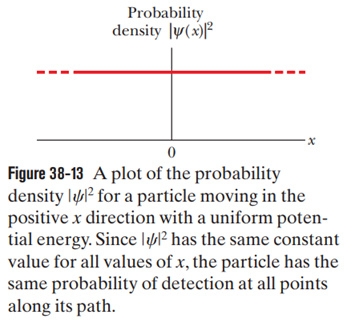
Suppose we put in Eq. 38-24 and relabeled as localid="1664290358337" .
(a) What would the resulting wave function then describe?
(b) How, if at all, would Fig. 38-13 be altered?
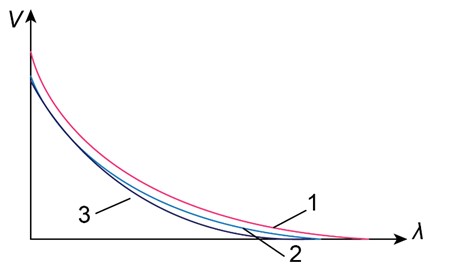
Photoelectric effect: Figure 38-20 gives the stopping voltage V versus the wavelength of light for three different materials. Rank the materials according to their work function, greatest first.
An orbiting satellite can charge by the photoelectric effect when sunlight ejects electrons from its outer surface. Satellites must be designed to minimize such charging because it can ruin the sensitive microelectronics. Suppose a satellite is coated with platinum, a metal with a very large function . Find the longest wavelength of incident sunlight that can eject an electron from the platinum.
An electron and a photon each have a wavelength of . What is the momentum (in ) of the (a) electron and (b) photon? What is the energy (in ) of the (c) electron and (d) photon?
What do you think about this solution?
We value your feedback to improve our textbook solutions.