Chapter 42: Q7Q (page 1301)
The nuclideis an alpha-emitter. Into which of the following nuclides does it decay:?
Short Answer
The nuclide will decay into through the alpha decay process.
Learning Materials
Features
Discover
Chapter 42: Q7Q (page 1301)
The nuclideis an alpha-emitter. Into which of the following nuclides does it decay:?
The nuclide will decay into through the alpha decay process.
All the tools & learning materials you need for study success - in one app.
Get started for free
A neutron star is a stellar object whose density is about that of nuclear matter, . Suppose that the Sun were to collapse and become such a star without losing any of its present mass. What would be its radius?
When an alpha particle collides elastically with a nucleus, the nucleus recoils. Suppose an 5.00MeV alpha particle has a head-on elastic collision with a gold nucleus that is initially at rest. What is the kinetic energy of (a) the recoiling nucleus and (b) the rebounding alpha particle?
The electric potential energy of a uniform sphere of charge qand radius ris given by (a) Does the energy represents a tendency for the sphere to bind together or blow apart? The nuclide is spherical with radius 6.64fm . For this nuclide, what are (b) the electric potential energy Uaccording to the equation, (c) the electric potential energy per proton, and (d) the electric potential energy per nucleon? The binding energy per nucleon is . (e) Why is the nuclide bound so well when the answers to (c) and (d) are large and positive?
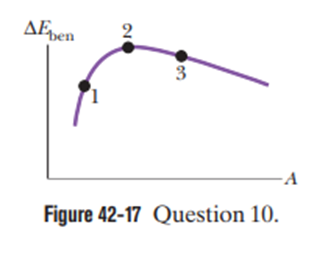
Figure 42-17 shows the curve for the binding energy per nucleonversus mass number A. Three isotopes are indicated. Rank them according to the energy required to remove a nucleon from the isotope, greatest first.
A rock recovered from far underground is found to contain 0.86 mg of , 0.15 mg of , and 1.6 mg of . How much will it likely contain? Assume that decays to only with a half-life of . Also assume that has a half-life of .
What do you think about this solution?
We value your feedback to improve our textbook solutions.