Chapter 42: Q10Q (page 1302)
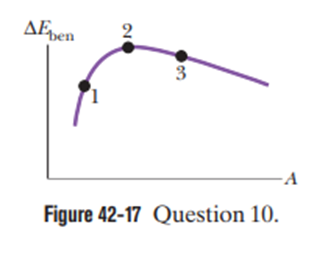
Figure 42-17 shows the curve for the binding energy per nucleonversus mass number A. Three isotopes are indicated. Rank them according to the energy required to remove a nucleon from the isotope, greatest first.
Short Answer
The rank of the points according to the energy required to remove a nucleon from the isotope is 2 > 3 > 1 .

