Chapter 39: Q69P (page 1218)
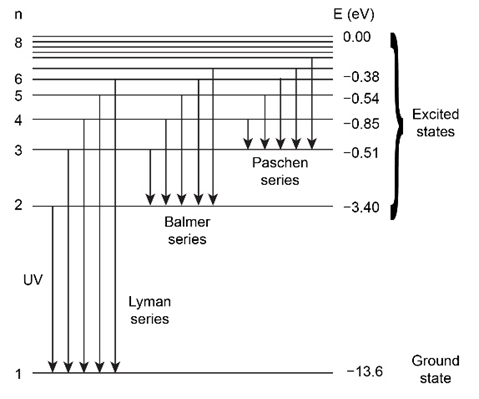
From the energy-level diagram for hydrogen, explain the observation that the frequency of the second Lyman-series line is the sum of the frequencies of the first Lyman-series line and the first Balmer-series line. This is an example of the empirically discovered Ritz combination principle. Use the diagram to find some other valid combinations.
Short Answer
It is shown that how the frequency of the second Lyman-series line is the sum of the frequencies of the first Lyman-series line and the first Balmer-series.
Another example of such transition is the transition associated with the second Balmer-series line can be thought of as (first Paschen) followed by (first Balmer).