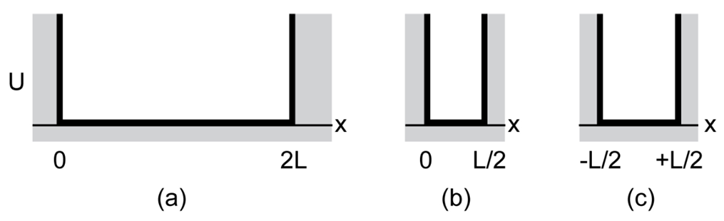
Chapter 39: Q36P (page 1216)
(a) What is the energy E of the hydrogen-atom electron whose probability density is represented by the dot plot of Fig. 39- 21? (b) What minimum energy is needed to remove this electron from the atom?
Short Answer
- The energy of the hydrogen-atom electron is .
- The required energy is .