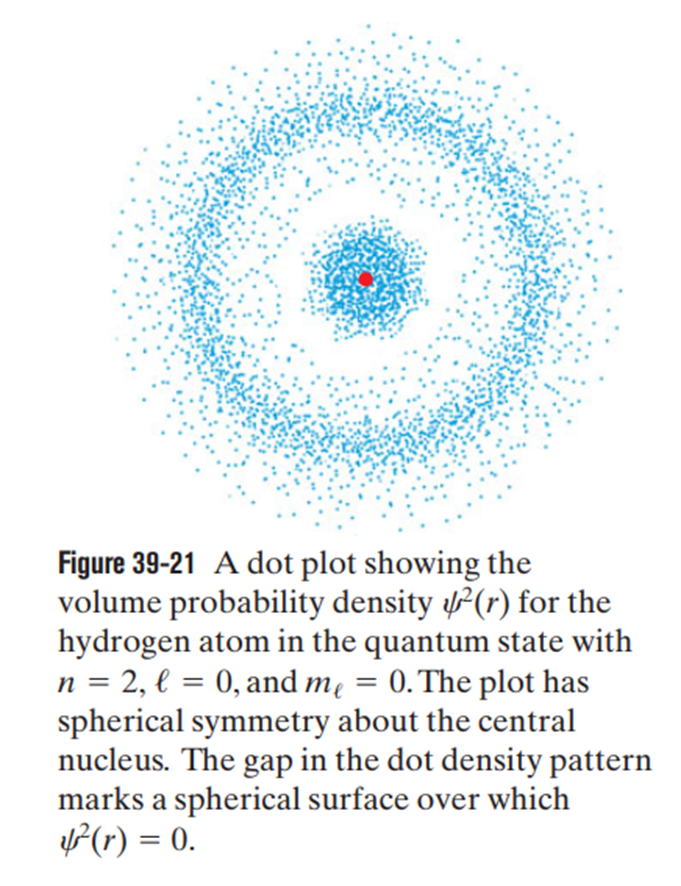
Chapter 39: Q19P (page 1215)
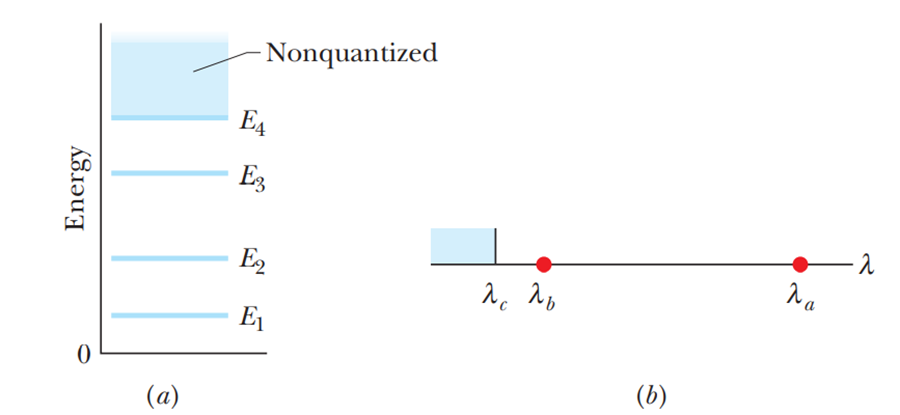
figure 39-28 shows the energy-level diagram for a finite, one-dimensional energy well that contains an electron. The nonquantized region begins at . Figure 39-28b gives the absorption spectrum of the electron when it is in the ground state—it can absorb at the indicated wavelengths: and for any wavelength less than . What is the energy of the first excited state?
Short Answer
The energy of the first excited state is 109 eV.