Chapter 39: Q10Q (page 1214)
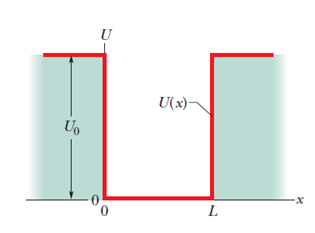
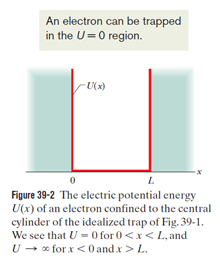
An electron, trapped in a finite potential energy well such as that of Fig. 39-7, is in its state of lowest energy. Are (a) its de-Broglie wavelength, (b) the magnitude of its momentum, and (c) its energy greater than, the same as, or less than they would be if the potential well were infinite, as in Fig. 39-2?
Short Answer
- The de-Broglie wavelength of the electron is greater than the finite well’s electron.
- Themagnitude of the momentum of the electron is less than the finite potential well’s electron.
- The energy of the electron trapped in an infinite potential well is less than that of the electron trapped in the finite potential well.