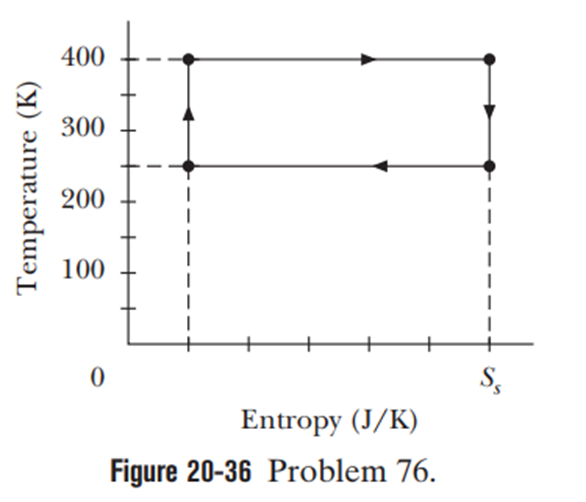
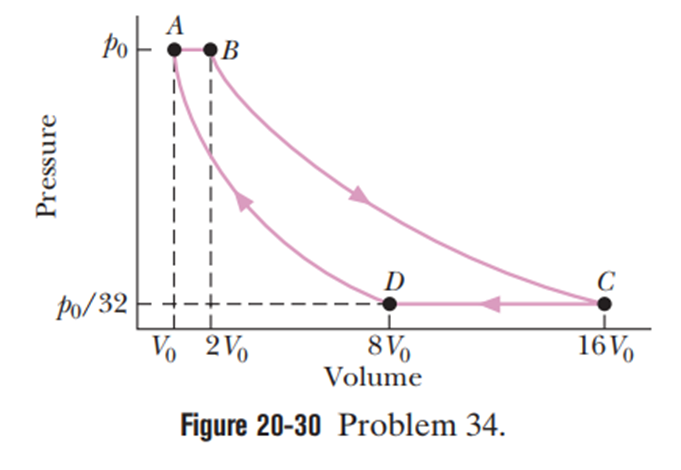
Chapter 20: Q34P (page 606)
An ideal gas ( 1.0 mol) is the working substance in an engine that operates on the cycle shown in Figure 20-30. Processes BC andDA are reversible and adiabatic. (a) Is the gas monatomic, diatomic, or polyatomic? (b) What is the engine efficiency?
Short Answer
- The gas is monatomic.
- The engine efficiency is 75%.