Chapter 43: Q6P (page 1330)
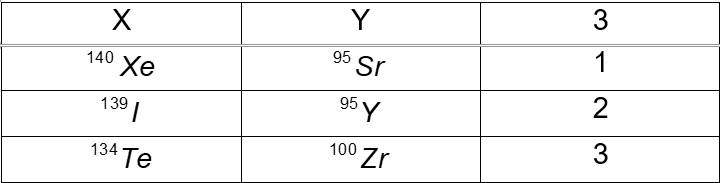
(a) – (d) Complete the following table, which refers to the generalized fission reaction

Short Answer
Learning Materials
Features
Discover
Chapter 43: Q6P (page 1330)
(a) – (d) Complete the following table, which refers to the generalized fission reaction


All the tools & learning materials you need for study success - in one app.
Get started for free
At the center of the Sun, the density of the gas is and the composition is essentially 35% hydrogen by mass and 65% helium by mass. (a) What is the number density of protons there? (b) What is the ratio of that proton density to the density of particles in an ideal gas at standard temperature (0°C) and pressure ?
Many fear that nuclear power reactor technology will increase the likelihood of nuclear war because reactors can be used not only to produce electrical energy but also, as a by-product through neutron capture with inexpensive , to make , which is a “fuel” for nuclear bombs. What simple series of reactions involving neutron capture and beta decay would yield this plutonium isotope?
The nuclide requires4.2 MeVfor fission. To remove a neutron from this nuclide requires an energy expenditure of 5.0 MeV. Isfissionable by thermal neutrons?
Calculate the energy released in the fission reaction
Here are some atomic and particle masses.
The effective Q for the proton–proton cycle of Fig. 43-11 is 26.2 MeV. (a) Express this as energy per kilogram of hydrogen consumed. (b) The power of the Sun is . If its energy derives from the proton–proton cycle, at what rate is it losing hydrogen? (c) At what rate is it losing mass? (d) Account for the difference in the results for (b) and (c). (e) The mass of the Sun is . If it loses mass at the constant rate calculated in (c), how long will it take to lose 0.10% of its mass?
What do you think about this solution?
We value your feedback to improve our textbook solutions.