Chapter 21: Q45P (page 628)
How many megacoulombs of positive charge are in of neutral molecular-hydrogen gas ()?
Short Answer
The value of the positive charge in the hydrogen molecule is.
Learning Materials
Features
Discover
Chapter 21: Q45P (page 628)
How many megacoulombs of positive charge are in of neutral molecular-hydrogen gas ()?
The value of the positive charge in the hydrogen molecule is.
All the tools & learning materials you need for study success - in one app.
Get started for free
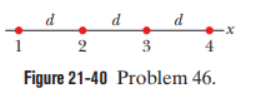
Question: In Fig. 21-40, four particles are fixed along anxaxis, separated bydistances -2.00. The charges are, with. In unit-vector notation, what is the net electrostatic force on (a) particle 1 and (b) particle 2 due to the other particles?
In a spherical metal shell of radius R, an electron is shot from the center directly toward a tiny hole in the shell, through which it escapes. The shell is negatively charged with a surface charge density (charge per unit area) of.What is the magnitude of the electron’s acceleration when it reaches radial distances (a)and (b)?
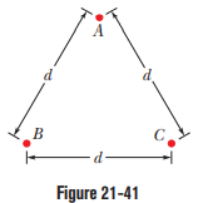
Question: In Fig. 21-41, three identical conducting spheres form an equilateral triangle of side length d=20.0 cm. The sphere radii are much smaller thand, and the sphere charges are,
(a) What is the magnitude of the electrostatic force between spheresAandC?
The following steps are then taken:AandBare connected by a thin wire andthen disconnected;Bis grounded by the wire, and the wire is then removed;BandCare connected by the wire and then disconnected. What now are the magnitudes of the electrostatic force (b) between spheresAandCand (c) between spheresBandC?
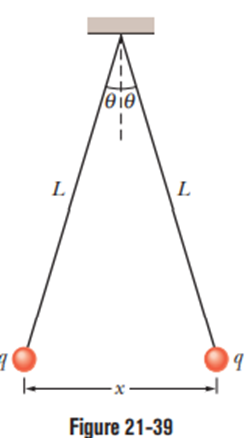
Question: (a) Explain what happens to the balls of Problem 42 if one of them is discharged (loses its charge qto, say, the ground). (b) Find the new equilibrium separation x, using the given values of Land mand the computed value of |q|.
The charges of an electron and a positron are . The mass of each is.What is the ratio of the electrical force to the gravitational force between an electron and a positron?
What do you think about this solution?
We value your feedback to improve our textbook solutions.