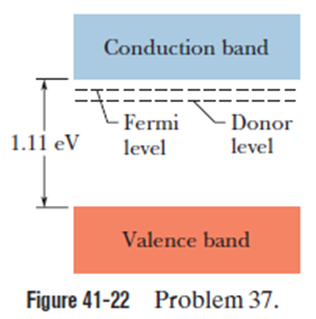
Chapter 41: Q42P (page 1275)
A potassium chloride crystal has an energy band gap of 7.6eV above the topmost occupied band, which is full. Is this crystal opaque or transparent to light of wavelength 140 nm?
Short Answer
The crystal is opaque to light of wavelength 140 nm .