Chapter 41: Q3P (page 1273)
Copper, a monovalent metal, has molar mass 63.54 g/mol and density . What is the number density nof conduction electrons in copper?
Short Answer
Conduction electrons, in copper, have a number density .
Learning Materials
Features
Discover
Chapter 41: Q3P (page 1273)
Copper, a monovalent metal, has molar mass 63.54 g/mol and density . What is the number density nof conduction electrons in copper?
Conduction electrons, in copper, have a number density .
All the tools & learning materials you need for study success - in one app.
Get started for free
(a) Show that the density of states at the Fermi energy is given by
in which nis the number density of conduction electrons.
(b) Calculate for copper, which is a monovalent metal with molar mass 63.54g/mol and density .
Verify your calculation with the curve of Fig. 41-6, recalling that =for copper.
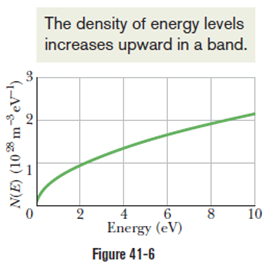
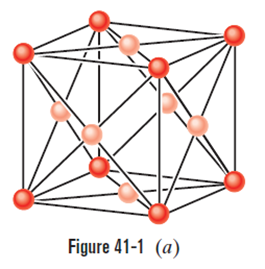
Figure 41-1ashows 14 atoms that represent the unit cell of copper. However, because each of these atoms is shared with one or more adjoining unit cells, only a fraction of each atom belongs to the unit cell shown. What is the number of atoms per unit cell for copper? (To answer, count up the fractional atoms belonging to a single unit cell.)
A sample of a certain metal has a volume of . The metal has a density of and a molar mass of 60 g/mol. The atoms are bivalent. How many conduction electrons (or valence electrons) are in the sample?
Calculate the density of occupied states, for copper at 10000K for energy Eof (a)4.00eV , (b) 6.75eV, (c) 7.00eV, (d) 7.25eV, and (e) 9.00eV. Compare your results with the graph of Fig. 41-8b.The Fermi energy for copper is 7.00eV.
A certain computer chip that is about the size of a postage stamp contains about 3.5 million transistors. If the transistors are square, what must be their maximumdimension? (Note:Devices other than transistors are also on the chip, and there must be room for the interconnections among the circuit elements. Transistors smaller than are now commonly and inexpensively fabricated.)
What do you think about this solution?
We value your feedback to improve our textbook solutions.