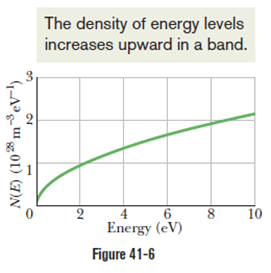
Chapter 41: Q2Q (page 1272)
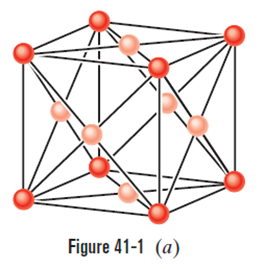
Figure 41-1ashows 14 atoms that represent the unit cell of copper. However, because each of these atoms is shared with one or more adjoining unit cells, only a fraction of each atom belongs to the unit cell shown. What is the number of atoms per unit cell for copper? (To answer, count up the fractional atoms belonging to a single unit cell.)
Short Answer
The number of atoms per unit cell is 4.