Chapter 40: Q9Q (page 1246)
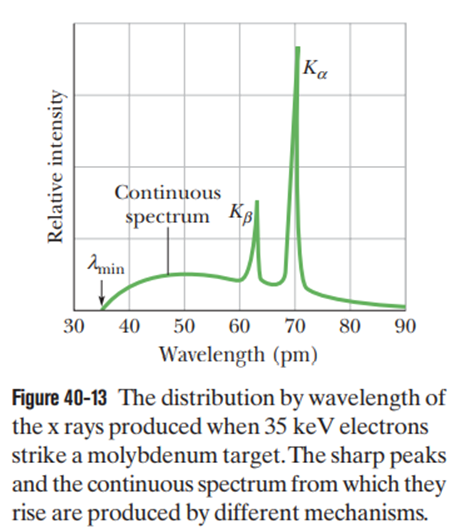
The x-ray line for any element arises because of a transition between the Kshell (n=1) and the Lshell (n=2). Figure 40-13 shows this line (for a molybdenum target) occurring at a single wavelength. With higher resolution, however, the line splits into several wavelength components because the Lshell does not have a unique energy.
(a) How many components does the line have?
(b) Similarly, how many components does the line have?
Short Answer
a) The line has components of a single wavelength.
b) The line has components of a single wavelength.


