Chapter 40: Q62P (page 1250)
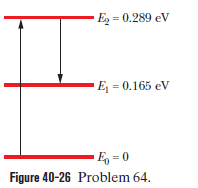
Ruby lasers are at a wavelength of 694 nm. A certain ruby crystal has Cr ions (which are the atoms that lase). The lasing transition is between the first excited state and the ground state, and the output is a light pulse lasting . As the pulse begins, 60.0% of the Cr ions are in the first excited state and the rest are in the ground state. What is the average power emitted during the pulse? (Hint:Don’t just ignore the ground-state ions.)
Short Answer
The average power emitted during the pulse is .