Chapter 40: Q2P (page 1247)
How many electron states are there in a shell defined by the quantum number n = 5 ?
Short Answer
There are 50 electron states in the shell defined by the quantum number n = 5 .
Learning Materials
Features
Discover
Chapter 40: Q2P (page 1247)
How many electron states are there in a shell defined by the quantum number n = 5 ?
There are 50 electron states in the shell defined by the quantum number n = 5 .
All the tools & learning materials you need for study success - in one app.
Get started for free
A rectangular corral of widths and contains seven electrons. What multiple of gives the energy of the ground state of the system? Assume that the electrons do not interact with one another, and do not neglect spin.
From Eq. 40-26, what is the ratio of the photon energies due to transitions in two atoms whose atomic numbers are Z and Z' ? (b) What is this ratio for uranium and aluminum? (c) For uranium and lithium?
On which quantum numbers does the energy of an electron depend in (a) a hydrogen atom and
(b) a vanadium atom?
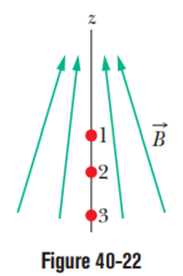
Figure 40-22 shows three points at which a spin-up electron can be placed in a non-uniform magnetic field (there is a gradient along the z-axis).
(a) Rank the three points according to the energy Uof the electron’s intrinsic magnetic dipole moment , most positive first.
(b) What is the direction of the force on the electron due to the magnetic field if the spin-up electron is at point 2?
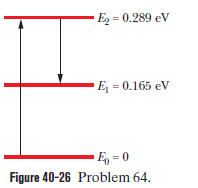
Martian laser. Where sunlight shines on the atmosphere of Mars, carbon dioxide molecules at an altitude of about 75 km undergo natural laser action. The energy levels involved in the action are shown in Fig. 40-26; population inversion occurs between energy levels and . (a) What wavelength of sunlight excites the molecules in the lasing action? (b) At what wavelength does lasing occur? (c) In what region of the electromagnetic spectrum do the excitation and lasing wavelengths lie?
What do you think about this solution?
We value your feedback to improve our textbook solutions.