Chapter 15: Q14PE (page 550)
Calculate the net work output of a heat engine following path ABCDA in the figure below.
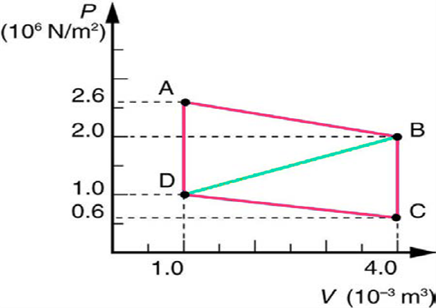
Short Answer
The net work output of this heat engine is 4800 J.
Learning Materials
Features
Discover
Chapter 15: Q14PE (page 550)
Calculate the net work output of a heat engine following path ABCDA in the figure below.

The net work output of this heat engine is 4800 J.
All the tools & learning materials you need for study success - in one app.
Get started for free
A\({\bf{4}}\)-ton air conditioner removes\({\bf{5}}.{\bf{06}} \times {\bf{1}}{{\bf{0}}^{\bf{7}}}\;{\bf{J}}\)(\({\bf{48}},{\bf{000}}\)British thermal units) from a cold environment in\({\bf{1}}.{\bf{00}}\;{\bf{h}}\). (a) What energy input in joules is necessary to do this if the air conditioner has an energy efficiency rating ( EER) of\({\bf{12}}.{\bf{0}}\)? (b) What is the cost of doing this if the work costs\({\bf{10}}.{\bf{0}}\)cents per\({\bf{3}}.{\bf{60}} \times {\bf{1}}{{\bf{0}}^{\bf{6}}}\;{\bf{J}}\)(one kilowatt-hour)? (c) Discuss whether this cost seems realistic. Note that the energy efficiency rating ( EER) of an air conditioner or refrigerator is defined to be the number of British thermal units of heat transfer from a cold environment per hour divided by the watts of power input.
Does a gas become more orderly when it liquefies? Does its entropy change? If so, does the entropy increase or decrease? Explain your answer.
Suppose you have an ideal refrigerator that cools an environment at −20.0ºC and has heat transfer to another environment at 50.0ºC . What is its coefficient of performance?
What is the change in entropy in an adiabatic process? Does this imply that adiabatic processes are reversible? Can a process be precisely adiabatic for a macroscopic system?
The Sun radiates energy at the rate of 3.80×1026 W from its 5500º C surface into dark empty space (a negligible fraction radiates onto Earth and the other planets). The effective temperature of deep space is −270º C . (a) What is the increase in entropy in one day due to this heat transfer? (b) How much work is made unavailable?
What do you think about this solution?
We value your feedback to improve our textbook solutions.