Chapter 21: Q14PE (page 775)
Standard automobile batteries have six lead-acid cells in series, creating a total emf of 12.0 V. What is the emf of an individual lead-acid cell?
Short Answer
The emf of an individual lead-acid cell is 2 V.
Learning Materials
Features
Discover
Chapter 21: Q14PE (page 775)
Standard automobile batteries have six lead-acid cells in series, creating a total emf of 12.0 V. What is the emf of an individual lead-acid cell?
The emf of an individual lead-acid cell is 2 V.
All the tools & learning materials you need for study success - in one app.
Get started for free
Why is the power dissipated by a closed switch, such as in Figure 21.43, small?
Show that if two resistors and are combined and one is much greater than the other : (a) Their series resistance is very nearly equal to the greater resistance (b) Their parallel resistance is very nearly equal to smaller resistance .
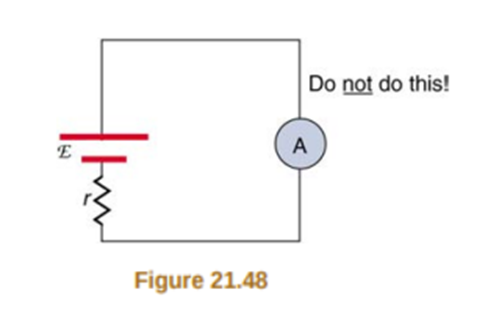
Why should you not connect an ammeter directly across a voltage source as shown in Figure21.48?(Note that script Ein the figure stands for emf.)
Explain which battery is doing the charging and which is being charged in Figure 21.45.
When an unknown resistanceis placed in a Wheatstone bridge, it is possible to balance the bridge by adjusting to be . What is if?
What do you think about this solution?
We value your feedback to improve our textbook solutions.