Chapter 30: Q12PE (page 1112)
A hydrogen atom in an excited state can be ionized with less energy than when it is in its ground state. What is n for a hydrogen atom if 0.850 eV of energy can ionize it?
Short Answer
The value of n is 4.
Learning Materials
Features
Discover
Chapter 30: Q12PE (page 1112)
A hydrogen atom in an excited state can be ionized with less energy than when it is in its ground state. What is n for a hydrogen atom if 0.850 eV of energy can ionize it?
The value of n is 4.
All the tools & learning materials you need for study success - in one app.
Get started for free
(a) How many electrons can be in the\(n{\rm{ }} = {\rm{ }}4\)shell? (b) What are its subshells, and how many electrons can be in each?
Observers at a safe distance from an atmospheric test of a nuclear bomb feel its heat but receive none of its copious x rays. Why is air opaque to x rays but transparent to infrared?
Consider the Doppler-shifted hydrogen spectrum received from a rapidly receding galaxy. Construct a problem in which you calculate the energies of selected spectral lines in the Balmer series and examine whether they can be described with a formula like that in the equation\[\frac{{\rm{1}}}{{\rm{\lambda }}}{\rm{ = R}}\left( {\frac{{\rm{1}}}{{{\rm{n}}_{\rm{f}}^{\rm{2}}}}{\rm{ - }}\frac{{\rm{1}}}{{{\rm{n}}_{\rm{i}}^{\rm{2}}}}} \right){\rm{,}}\]but with a different constant R.
Why does the energy of characteristic x rays become increasingly greater for heavier atoms?
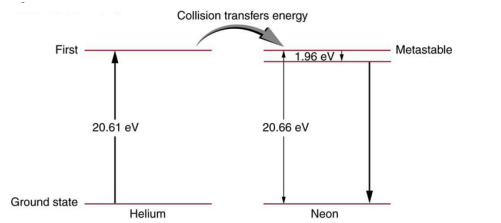
Figure 30.39 shows the energy-level diagram for neon.
(a) Verify that the energy of the photon emitted when neon goes from its metastable state to the one immediately below is equal to 1.96 eV.
(b) Show that the wavelength of this radiation is 633 nm.
(c) What wavelength is emitted when the neon makes a direct transition to its ground state?
What do you think about this solution?
We value your feedback to improve our textbook solutions.