Chapter 7: Q. 7.75P. (page 326)
Consider a gas of noninteracting spin-0 bosons at high temperatures, when . (Note that “high” in this sense can still mean below 1 K.)
- Show that, in this limit, the Bose-Einstein function can be written approximately as
. - Keeping only the terms shown above, plug this result into equation 7.122 to derive the first quantum correction to the chemical potential for gas of bosons.
- Use the properties of the grand free energy (Problems 5.23 and 7.7) to show that the pressure of any system is given by In , where is the grand partition function. Argue that, for gas of noninteracting particles, In can be computed as the sum over all modes (or single-particle states) of In , where ; is the grand partition function for the mode.
- Continuing with the result of part (c), write the sum over modes as an integral over energy, using the density of states. Evaluate this integral explicitly for gas of noninteracting bosons in the high-temperature limit, using the result of part (b) for the chemical potential and expanding the logarithm as appropriate. When the smoke clears, you should find
,
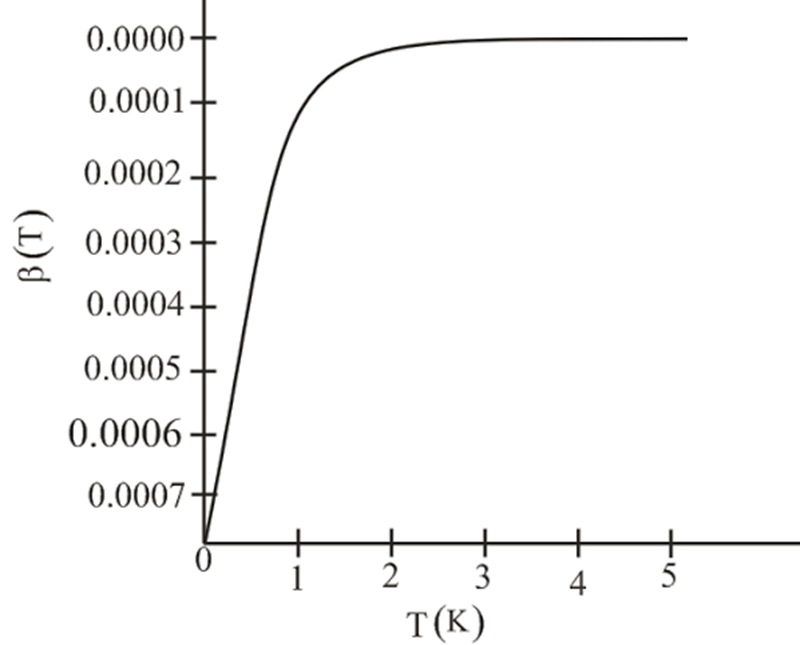
again neglecting higher-order terms. Thus, quantum statistics results in a lowering of the pressure of a boson gas, as one might expect. - Write the result of part (d) in the form of the virial expansion introduced in Problem 1.17, and read off the second virial coefficient, . Plot the predicted for a hypothetical gas of noninteracting helium-4 atoms.
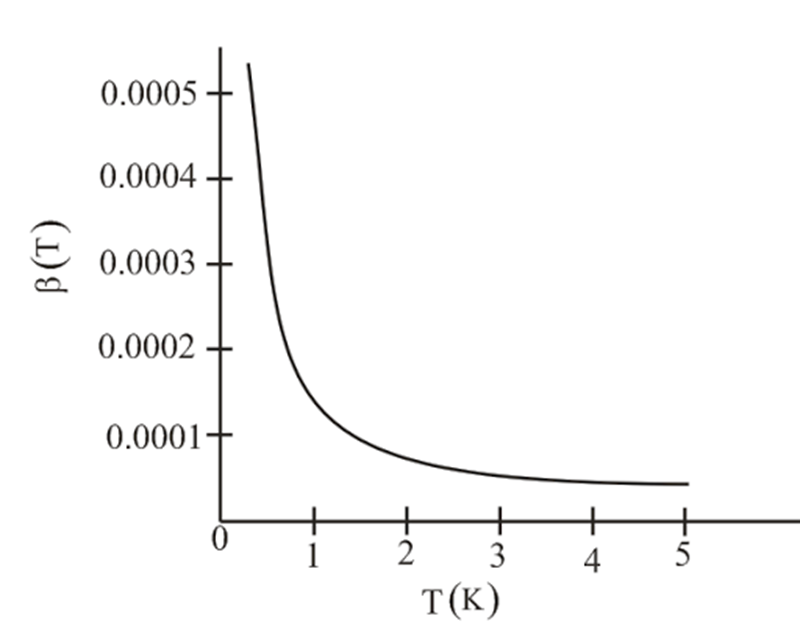
- Repeat this entire problem for gas of spin-1/2 fermions. (Very few modifications are necessary.) Discuss the results, and plot the predicted virial coefficient for a hypothetical gas of noninteracting helium-3 atoms.
Short Answer
- The Bose-Einstein distribution is given as .
- The chemical potential is .
- The pressure of a gas is given as and the logarithm of the grand partition function is computed as a sum over all modes.
- The pressure of a gas is .
- The value of the virial coefficient for helium is given as .
- For spin, half particle number density is given as . The viral coefficient is given as and the value of the virial coefficient is given as .