Chapter 7: Q. 7.51. (page 303)
The tungsten filament of an incandescent light bulb has a temperature of approximately . The emissivity of tungsten is approximately , and you may assume that it is independent of wavelength.
(a) If the bulb gives off a total of watts, what is the surface area of its filament in square millimetres?
(b) At what value of the photon energy does the peak in the bulb's spectrum occur? What is the wavelength corresponding to this photon energy?
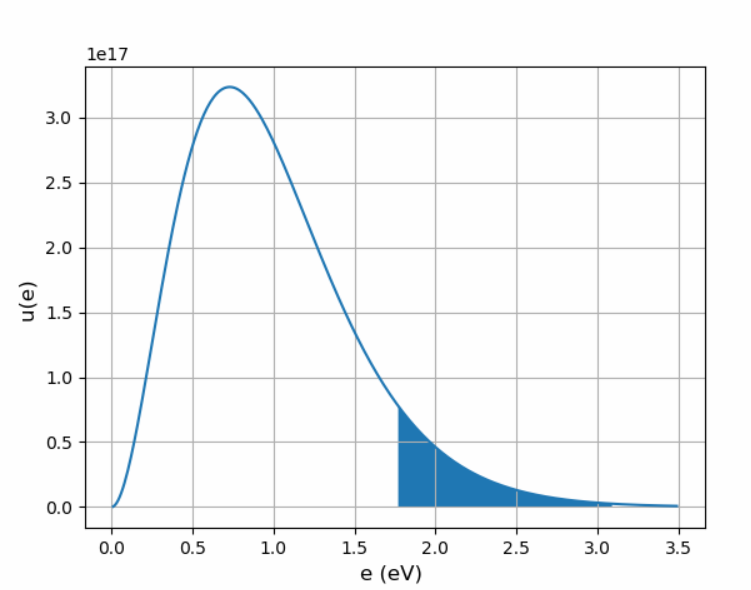
(c) Sketch (or use a computer to plot) the spectrum of light given off by the filament. Indicate the region on the graph that corresponds to visible wavelengths, between.
(d) Calculate the fraction of the bulb's energy that comes out as visible light. (Do the integral numerically on a calculator or computer.) Check your result qualitatively from the graph of part (c).
( e) To increase the efficiency of an incandescent bulb, would you want to raise or lower the temperature? (Some incandescent bulbs do attain slightly higher efficiency by using a different temperature.)
(f) Estimate the maximum possible efficiency (i.e., fraction of energy in the visible spectrum) of an incandescent bulb, and the corresponding filament temperature. Neglect the fact that tungsten melts at .
Short Answer
(a) The surface area of its filament is .
(b) The wavelength corresponding to this photon energy is.
(c) The spectrum of the light given off is
(d) The fraction of the bulb's energy that comes out as visible light is.
(e) To increase the efficiency of the incandescent bulb we will raise the temperature.
(f) The maximum possible efficiency is achieved at temperature.