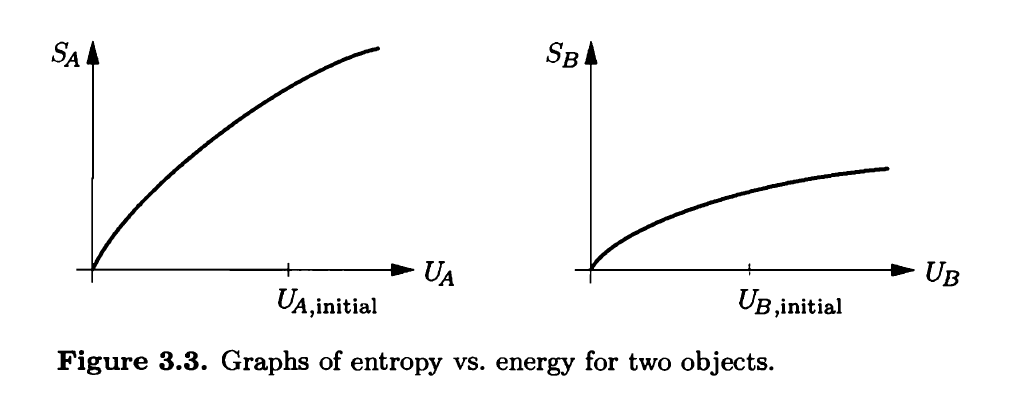
Chapter 3: Q. 3.31 (page 114)
Experimental measurements of heat capacities are often represented in reference works as empirical formulas. For graphite, a formula that works well over a fairly wide range of temperatures is (for one mole)
where , and . Suppose, then, that a mole of graphite is heated at constant pressure from to . Calculate the increase in its entropy during this process. Add on the tabulated value of (from the back of this book) to obtain .