Chapter 3: Q. 3.15 (page 97)
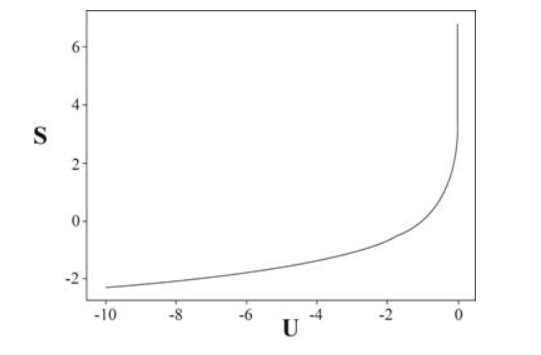
In Problem 1.55 you used the virial theorem to estimate the heat capacity of a star. Starting with that result, calculate the entropy of a star, first in terms of its average temperature and then in terms of its total energy. Sketch the entropy as a function of energy, and comment on the shape of the graph.
Short Answer
The required expression for the entropy of a star is and the graph can be sketched as below.