Chapter 3: Q. 3.19 (page 107)
Fill in the missing algebraic steps to derive equations 3.30, 3.31, and 3.33.
Short Answer
Thus the equations are derived to fill the missing steps.
Learning Materials
Features
Discover
Chapter 3: Q. 3.19 (page 107)
Fill in the missing algebraic steps to derive equations 3.30, 3.31, and 3.33.
Thus the equations are derived to fill the missing steps.
All the tools & learning materials you need for study success - in one app.
Get started for free
A bit of computer memory is some physical object that can be in two different states, often interpreted as 0 and 1. A byte is eight bits, a kilobyte is bytes, a megabyte is 1024 kilobytes, and a gigabyte is 1024 megabytes.
(a) Suppose that your computer erases or overwrites one gigabyte of memory, keeping no record of the information that was stored. Explain why this process must create a certain minimum amount of entropy, and calculate how much.
(b) If this entropy is dumped into an environment at room temperature, how much heat must come along with it? Is this amount of heat significant?
Show that the entropy of a two-state paramagnet, expressed as a function of temperature, is , where . Check that this formula has the expected behavior as and .
Polymers, like rubber, are made of very long molecules, usually tangled up in a configuration that has lots of entropy. As a very crude model of a rubber band, consider a chain of N links, each of length (see Figure 3.17). Imagine that each link has only two possible states, pointing either left or right. The total length L of the rubber band is the net displacement from the beginning of the first link to the end of the last link.
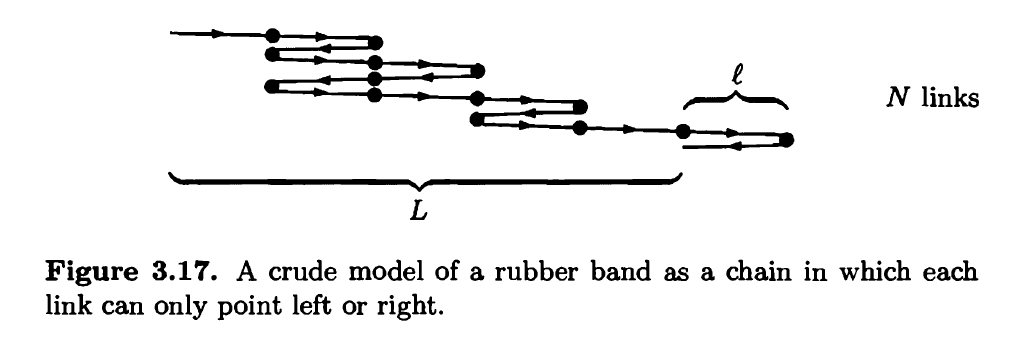
(a) Find an expression for the entropy of this system in terms of N and NR, the number of links pointing to the right.
(b) Write down a formula for L in terms of N and NR.
(c) For a one-dimensional system such as this, the length L is analogous to the volume V of a three-dimensional system. Similarly, the pressure P is replaced by the tension force F. Taking F to be positive when the rubber band is pulling inward, write down and explain the appropriate thermodynamic identity for this system.
(d) Using the thermodynamic identity, you can now express the tension force F in terms of a partial derivative of the entropy. From this expression, compute the tension in terms of L, T, N, and .
(e) Show that when L << N, the tension force is directly proportional to L (Hooke's law).
(f) Discuss the dependence of the tension force on temperature. If you increase the temperature of a rubber band, does it tend to expand or contract? Does this behavior make sense?
(g) Suppose that you hold a relaxed rubber band in both hands and suddenly stretch it. Would you expect its temperature to increase or decrease? Explain. Test your prediction with a real rubber band (preferably a fairly heavy one with lots of stretch), using your lips or forehead as a thermometer. (Hint: The entropy you computed in part (a) is not the total entropy of the rubber band. There is additional entropy associated with the vibrational energy of the molecules; this entropy depends on U but is approximately independent of L.)
Use a computer to reproduce Table 3.2 and the associated graphs of entropy, temperature, heat capacity, and magnetization. (The graphs in this section are actually drawn from the analytic formulas derived below, so your numerical graphs won't be quite as smooth.)
Experimental measurements of heat capacities are often represented in reference works as empirical formulas. For graphite, a formula that works well over a fairly wide range of temperatures is (for one mole)
where , and . Suppose, then, that a mole of graphite is heated at constant pressure from to . Calculate the increase in its entropy during this process. Add on the tabulated value of (from the back of this book) to obtain .
What do you think about this solution?
We value your feedback to improve our textbook solutions.