Chapter 5: Q 5.92 (page 219)
Suppose you have a box of atomic hydrogen, initially at room temperature and atmospheric pressure. You then raise the temperature, keeping the volume fixed.
(a) Find an expression for the fraction of the hydrogen that is ionised as a function of temperature. (You'll have to solve a quadratic equation.) Check that your expression has the expected behaviour at very low and very high temperatures.
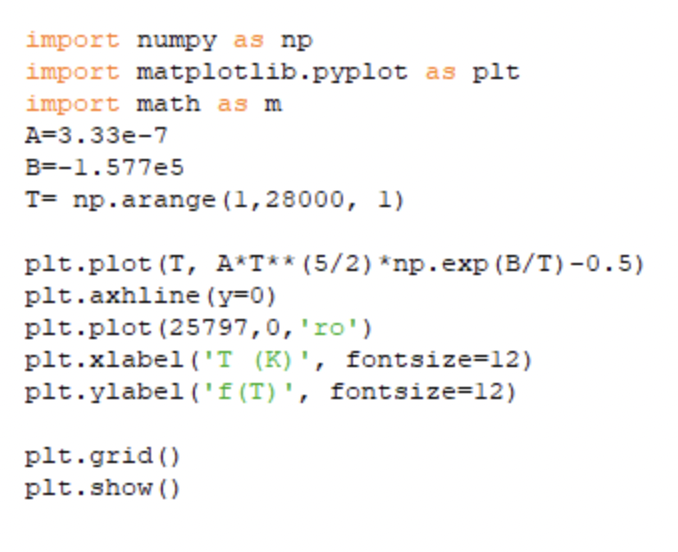
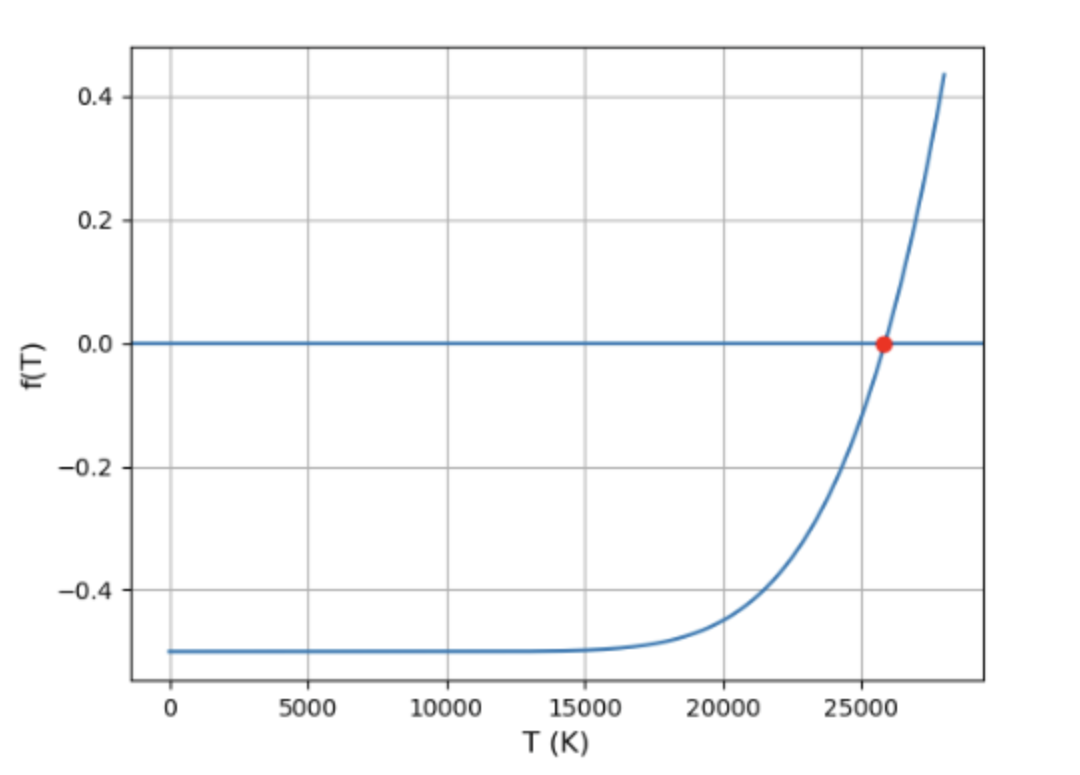
(b) At what temperature is exactly half of the hydrogen ionised?
(c) Would raising the initial pressure cause the temperature you found in part (b) to increase or decrease? Explain.
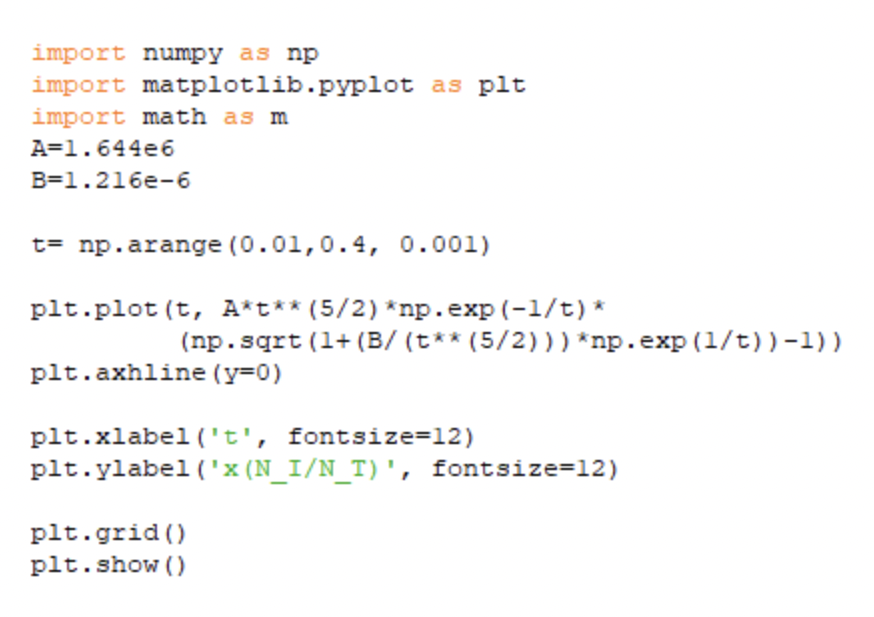
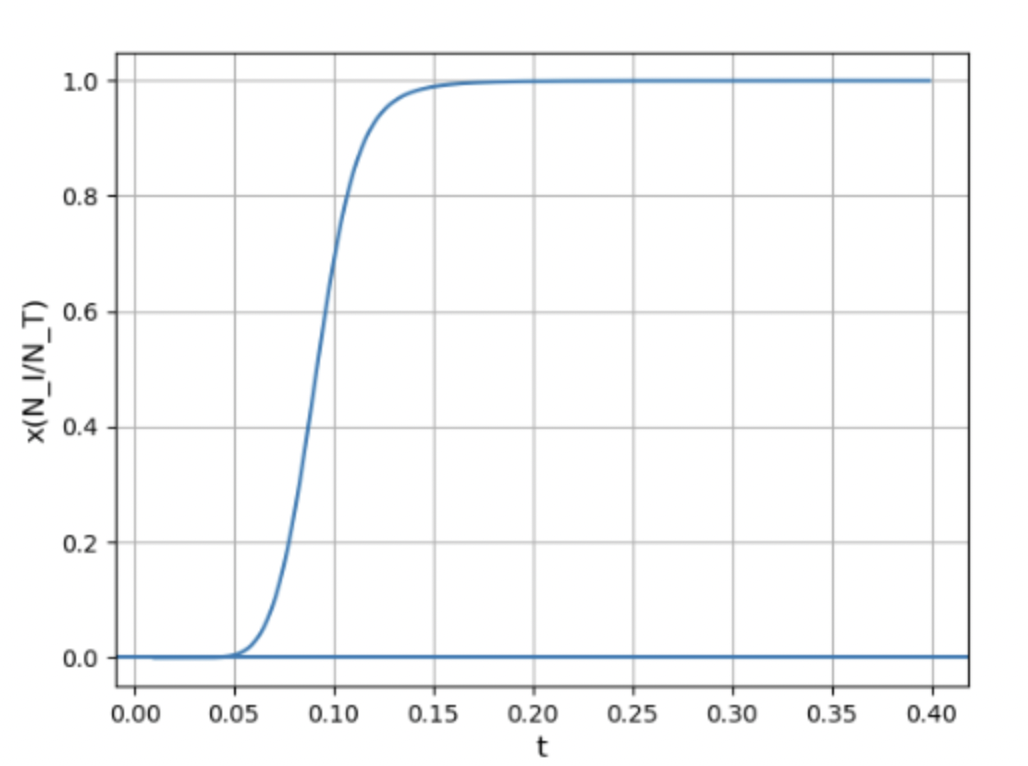
(d) Plot the expression you found in part (a) as a function of the dimension- less variable t = kT/I. Choose the range of t values to clearly show the interesting part of the graph.
Short Answer
(a) An expression for the fraction of the hydrogen that is ionised as a function temperature is
(b) The temperature at which exactly half of the hydrogen is ionised is T=25800K