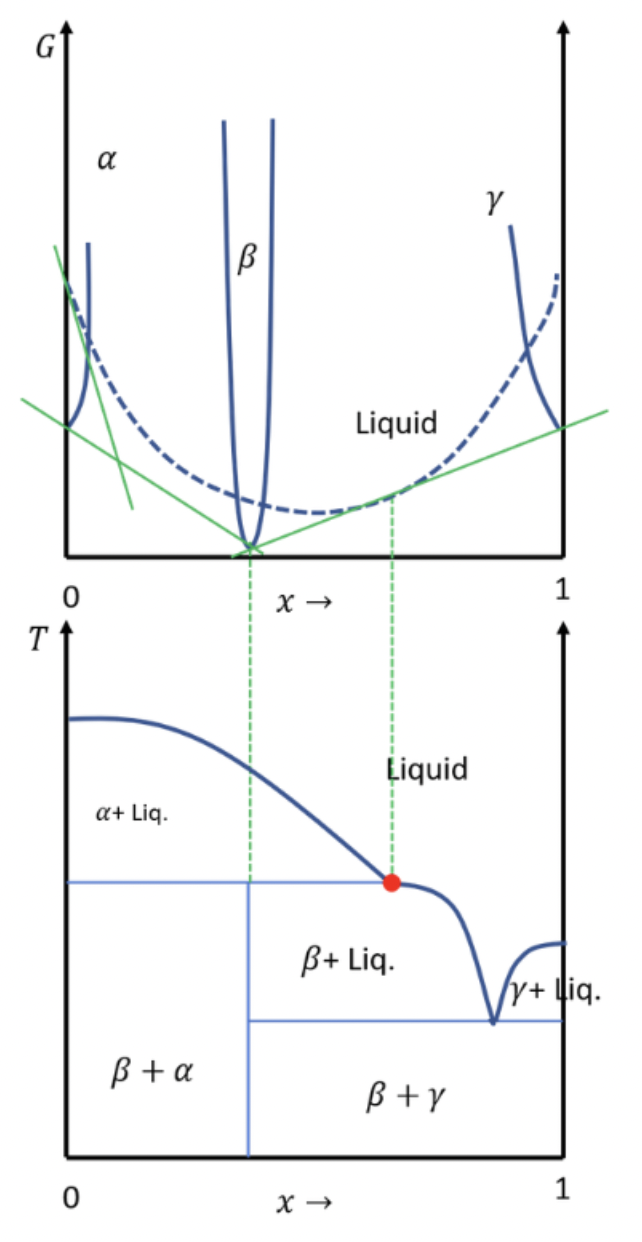
Chapter 5: Q 5.72 (page 200)
Repeat the previous problem for the diagram in Figure 5.35 (right), which has an important qualitative difference. In this phase diagram, you should find that and liquid are in equilibrium only at temperatures below the point where the liquid is in equilibrium with infinitesimal amounts of and . This point is called a peritectic point. Examples of systems with this behaviour include water + NaCl and leucite + quartz.
Short Answer
Starting at x=0 on the left, thephase and the liquid phase are stable, then the liquid phase is stable, then the phase and the liquid phase are stable, then a narrow range of x where only the phase is stable, and ultimately just the liquid phase is stable.