Chapter 5: Q. 5.77 (page 205)
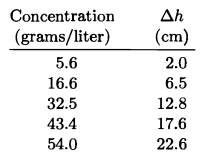
Osmotic pressure measurements can be used to determine the molecular weights of large molecules such as proteins. For a solution of large molecules to qualify as "dilute," its molar concentration must be very low and hence the osmotic pressure can be too small to measure accurately. For this reason, the usual procedure is to measure the osmotic pressure at a variety of concentrations, then extrapolate the results to the limit of zero concentration. Here are some data for the protein hemoglobin dissolved in water at :
| Concentration (grams/liter) | (cm) |
| 5.6 | 2.0 |
| 16.6 | 6.5 |
| 32.5 | 12.8 |
| 43.4 | 17.6 |
| 54.0 | 22.6 |
The quantity is the equilibrium difference in fluid level between the solution and the pure solvent,. From these measurements, determine the approximate molecular weight of hemoglobin (in grams per mole).
An experimental arrangement for measuring osmotic pressure. Solvent flows across the membrane from left to right until the difference in fluid level,, is just enough to supply the osmotic pressure.
Short Answer
The approximate molecular weight is .