Chapter 5: Q 5.8 (page 158)
Derive the thermodynamic identity for (equation 5.23), and from it the three partial derivative relations 5.24.
Short Answer
The expression for change in G is and the relations are
Learning Materials
Features
Discover
Chapter 5: Q 5.8 (page 158)
Derive the thermodynamic identity for (equation 5.23), and from it the three partial derivative relations 5.24.
The expression for change in G is and the relations are
All the tools & learning materials you need for study success - in one app.
Get started for free
Plot the Van der Waals isotherm for T/Tc = 0.95, working in terms of reduced variables. Perform the Maxwell construction (either graphically or numerically) to obtain the vapor pressure. Then plot the Gibbs free energy (in units of NkTc) as a function of pressure for this same temperature and check that this graph predicts the same value for the vapor pressure.
Consider the production of ammonia from nitrogen and hydrogen,
at 298 K and 1 bar. From the values of ΔH and S tabulated at the back of this book, compute ΔG for this reaction and check that it is consistent with the value given in the table.
Consider a fuel cell that uses methane ("natural gas") as fuel. The reaction is
(a) Use the data at the back of this book to determine the values of and for this reaction, for one mole of methane. Assume that the reaction takes place at room temperature and atmospheric pressure.
(b) Assuming ideal performance, how much electrical work can you get out of the cell, for each mole of methane fuel?
(c) How much waste heat is produced, for each mole of methane fuel?
(d) The steps of this reaction are
What is the voltage of the cell?
Use the Clausius-Clapeyron relation to derive equation 5.90 directly from Raoult's law. Be sure to explain the logic carefully.
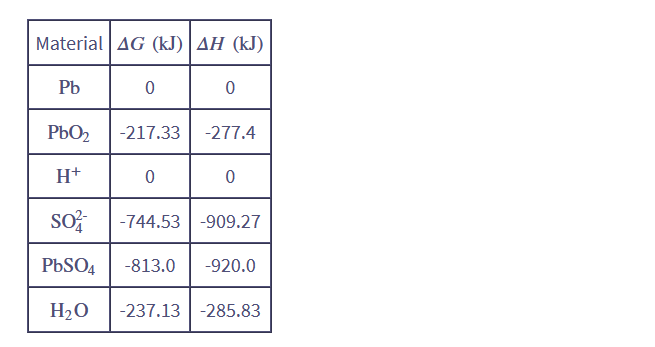
Use the data at the back of this book to verify the values of ΔH and ΔGquoted above for the lead-acid reaction 5.13.
At temp 298K and pressure 1 bar.
What do you think about this solution?
We value your feedback to improve our textbook solutions.