Chapter 5: Q 5.64 (page 195)
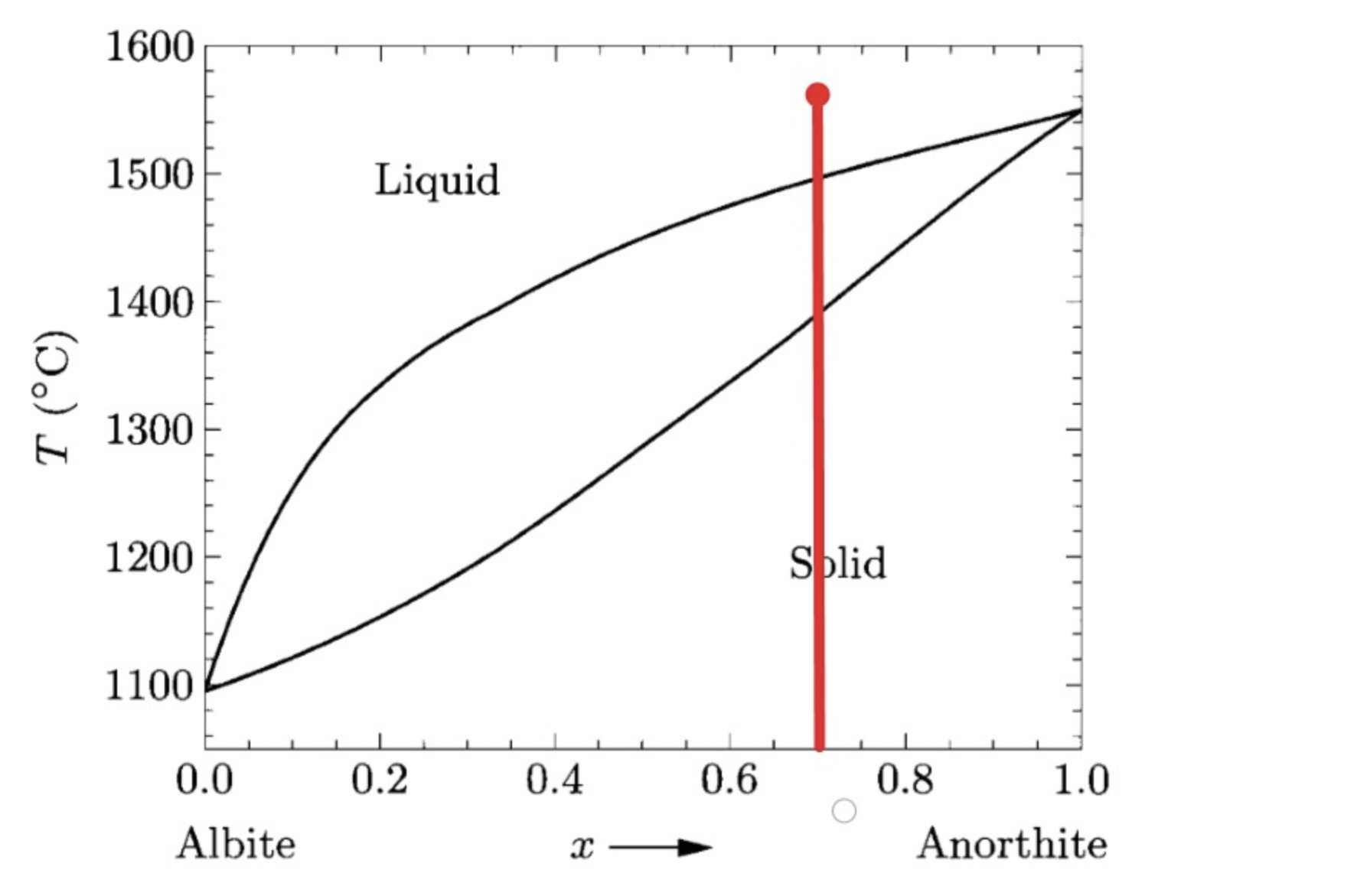
Problem 5.64. Figure 5.32 shows the phase diagram of plagioclase feldspar, which can be considered a mixture of albite and anorthite
a) Suppose you discover a rock in which each plagioclase crystal varies in composition from center to edge, with the centers of the largest crystals composed of 70% anorthite and the outermost parts of all crystals made of essentially pure albite. Explain in some detail how this variation might arise. What was the composition of the liquid magma from which the rock formed?
(b) Suppose you discover another rock body in which the crystals near the top are albite-rich while the crystals near the bottom are anorthite-rich. Explain how this variation might arise.
Short Answer
A)
B) The first anorthite-rich crystal forms, then the crystals are separated. The liquid portion of the composition will sink to the bottom, forming anorthite-rich crystals. Albite crystals will form at the very top.