Temperature diagram for mixture of nitrogen and oxygen
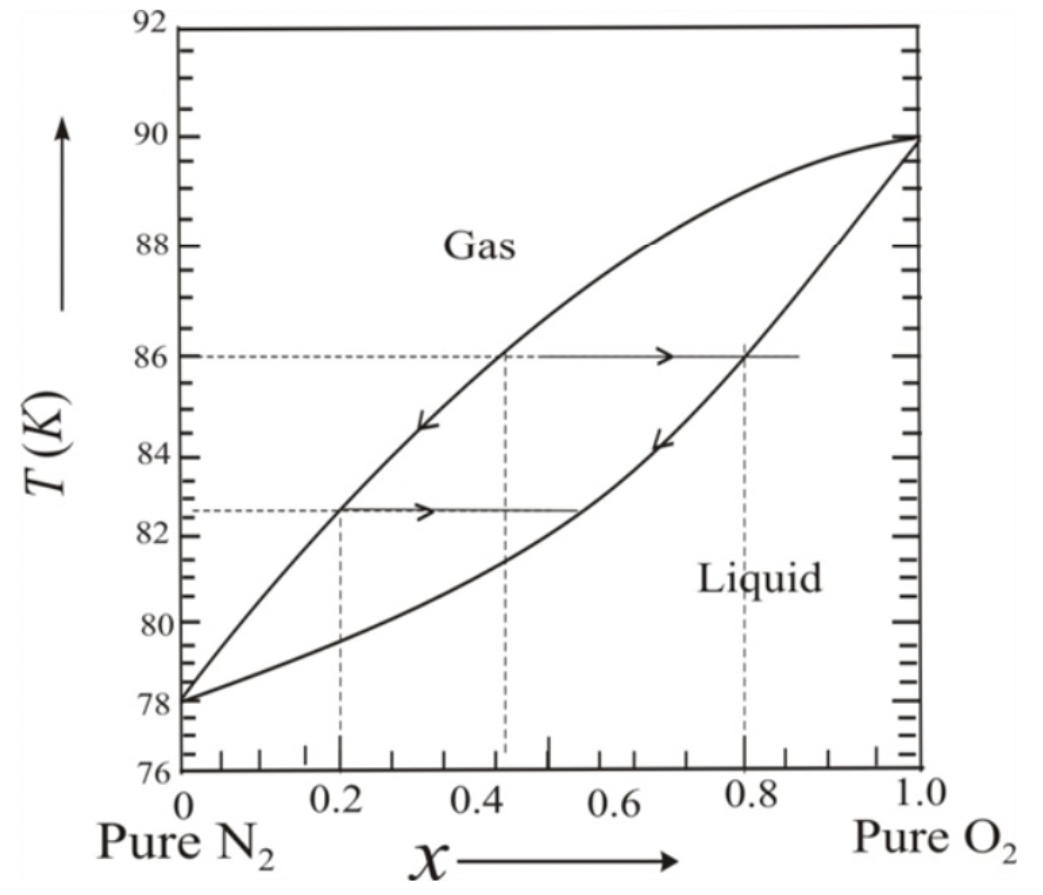
Consider the experimental phase diagram for nitrogen and oxygen at atmospheric pressure in Figure 5.31 of the book. Assume we have a mixture of 40% oxygen and 60% nitrogen, so x=0.4. As the temperature rises, the mixture remains in a liquid state until we reach 80.6 K, at which point the liquid begins to vaporise. If we draw a horizontal line from the lower curve to the upper curve (until they intersect), we can see that x= 0.16 from the upper curve at this temperature, and since pure nitrogen occurs at x = 0, we can conclude the vaporising process will continue as the temperature rises; follow the upper curve until it hits x = 0.40; there can't be any liquid left from oxygen at this point; drawing a horizontal line, we can see that the temperature at this point is 84.5 K.


