Chapter 5: Q 5.42 (page 177)
Ordinarily, the partial pressure of water vapour in the air is less than the equilibrium vapour pressure at the ambient temperature; this is why a cup of water will spontaneously evaporate. The ratio of the partial pressure of water vapour to the equilibrium vapour pressure is called the relative humidity. When the relative humidity is 100%, so that water vapour in the atmosphere would be in diffusive equilibrium with a cup of liquid water, we say that the air is saturated. The dew point is the temperature at which the relative humidity would be 100%, for a given partial pressure of water vapour.
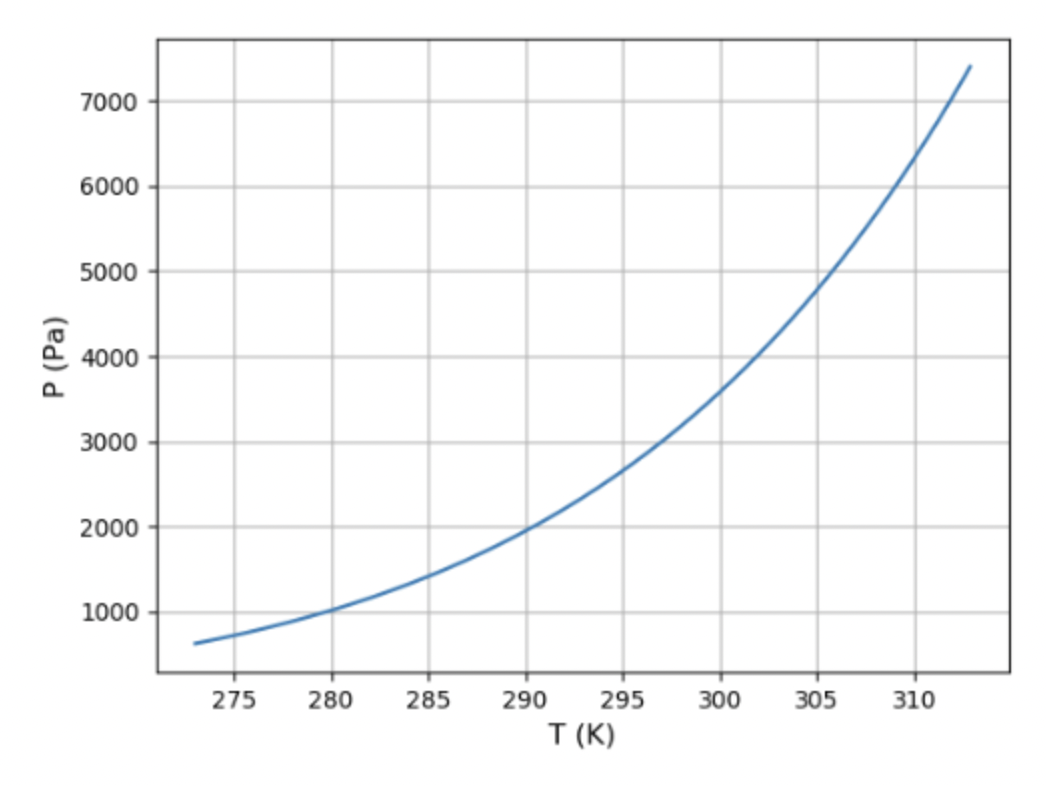
(a) Use the vapour pressure equation (Problem 5.35) and the data in Figure 5.11 to plot a graph of the vapour pressure of water from 0°C to 40°C. Notice that the vapour pressure approximately doubles for every 10° increase in temperature.
(b) Suppose that the temperature on a certain summer day is 30° C. What is the dew point if the relative humidity is 90%? What if the relative humidity is 40%?
Short Answer
When the relative humidity is 90%, the dew point is 28°C.
When the relative humidity is 40%, the dew point is 14.9°C.